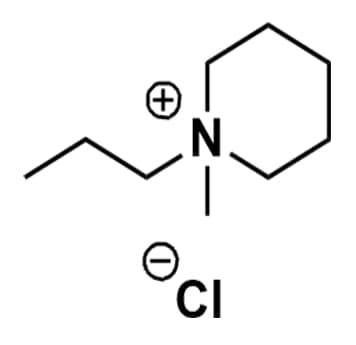
1-Methyl-1-propylpiperidinium chloride, >99%
Price range: $299.99 through $6,941.32
Product Code: IL-0151-HPCAS NO: 1383436-85-8
- Chemical Formula: C9H20ClN
- Synonyms: N-Methyl-N-propylpiperidinium chloride, PMPip Cl, PIP13 Cl
SUM Formula: C9H20ClN
Molecular Weight: 177.72
Melting Point: >280 °C
Density: 1.024 g/cm³
Purity: >99%
- SUM Formula: C9H20ClN
- Molecular Weight: 177.72
- Melting Point: >280 °C
- Density: 1.024 g/cm³
1-Methyl-1-propylpiperidinium chloride, CAS: 1383436-85-8
Key Applications:
Electrochemical Systems & Ionic Conductivity Modifiers
- Serves as a high‑purity quaternary ammonium salt for tailoring ionic strength and transport properties in non‑aqueous and mixed‑solvent electrolytes.
- Useful in electrodeposition, electro‑organic synthesis, and electrochemical separations where chloride coordination and cation size influence reaction pathways.
- Supports formulation of prototype ionic liquid electrolytes for early‑stage battery and capacitor research.
Solvent & Phase‑Behavior Engineering
- Functions as a hydrophilic ionic liquid precursor, enabling studies on solvation, ion pairing, and micro‑heterogeneity in polar media.
- Applied in liquid–liquid extraction, phase transfer catalysis, and reaction media optimization where chloride counterions enhance substrate transport.
Materials Science & Polymer Processing
- Acts as a structure‑directing additive in polymer blends, ion‑conducting membranes, and hybrid organic–inorganic materials.
- Supports research on ionic plasticization, thermal stability tuning, and surface charge modification in specialty polymers.
Surfactant, Colloid, and Interface Studies
- Used as a cationic surfactant analogue for probing micelle formation, colloidal stability, and interfacial charge density.
- Enables controlled studies of electrostatic assembly, nanoparticle dispersion, and thin‑film deposition.
Catalysis & Reaction Optimization
- Provides a tunable ionic environment for transition‑metal catalysis, organocatalysis, and chloride‑mediated activation pathways.
- Supports mechanistic studies where cation size, ring rigidity, and chloride coordination influence catalytic turnover.
Analytical & Spectroscopic Applications
- Serves as a reference ionic species for NMR, IR, and conductivity studies examining ion pairing, solvation shells, and dynamic exchange.
- Useful in chromatographic method development where controlled ionic strength improves separation performance.
Please contact us if you want to learn more or need assistance with your order.