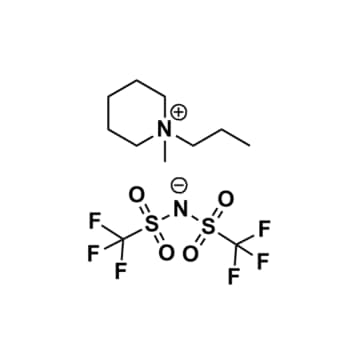
1-Methyl-1-propylpiperidinium bis(trifluoromethylsulfonyl)imide, >99.5%
Price range: $178.24 through $12,155.95
Product Code: IL-0045-UPCAS NO: 608140-12-1
- Chemical Formula: C11H20F6N2O4S2
- Synonyms: N-Methyl-N-propylpiperidinium bis(trifluoromethylsulfonyl)imide, PMPip BTA, PMPip NTf2, PMPip TFSI, PMPip BTI, PIP13 TFSI
- Weakly-coordinating anion
- Hydrophobic
For High Purity of >99% refer to IL-0045-HP
Conductivity: 2.12 mS/cm (30 °C)
SUM Formula: C11H20F6N2O4S2
Molecular Weight: 422.21
Melting Point: 8 °C
Density: 1.41 g/cm³ (23 °C)
ECW: 5.9 V
Purity: >99.5%
Viscosity: 176 cP (25 °C)
- SUM Formula: C11H20F6N2O4S2
- Molecular Weight: 422.21
- Melting Point: 8 °C
- Density: 1.41 g/cm³ (23 °C)
- ECW: 5.9 V
- Viscosity: 176 cP (25 °C)
1-Methyl-1-propylpiperidinium bis(trifluoromethylsulfonyl)imide, CAS: 608140-12-1
Key Applications:
Electrochemical Systems
- Serves as a non‑volatile electrolyte component for high‑temperature lithium and sodium battery research.
- Used as an ionically conductive medium in redox‑flow and hybrid flow battery development where oxidative stability is required.
- Functions as a supporting electrolyte in electrosynthesis, electrodeposition, and high‑potential anodic processes.
- Enables stable ionic environments for supercapacitors and double‑layer capacitor prototypes.
Materials Science and Polymer Processing
- Acts as a plasticizer or ionic mobility enhancer in polymer electrolytes, ionogels, and block‑copolymer membranes.
- Supports fabrication of high‑temperature, low‑flammability polymer systems due to its negligible vapor pressure and thermal resilience.
- Used in templating and dispersion of nanomaterials, including carbon nanostructures and metal oxides, where hydrophobic ionic liquids improve colloidal stability.
Catalysis and Synthesis
- Provides a stable, inert medium for transition‑metal catalysis, especially reactions requiring moisture‑free, high‑temperature conditions.
- Employed in biphasic catalysis where phase separation between hydrophobic ionic liquids and polar reactants improves catalyst recovery.
- Useful in electro‑organic synthesis requiring wide electrochemical windows and low nucleophilicity.
Separation Science
- Applied in liquid–liquid extraction systems for selective separation of metal ions, hydrophobic organics, and fluorinated species.
- Utilized in gas absorption studies, including CO₂ solubility and transport measurements in hydrophobic ionic liquids.
Thermal and Transport Studies
- Serves as a benchmark ionic liquid for evaluating ion transport, viscosity–conductivity relationships, and high‑temperature stability in piperidinium‑based systems.
- Used in fundamental studies comparing saturated versus unsaturated cation families (piperidinium vs. imidazolium/pyrrolidinium).
Please contact us if you want to learn more or need assistance with your order.