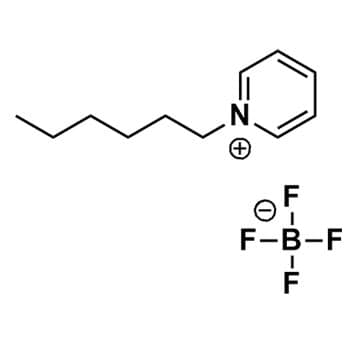
1-Hexylpyridinium tetrafluoroborate, >99%
Price range: $236.34 through $10,209.70
Product Code: IL-0108-HPCAS NO: 474368-70-2
- Chemical Formula: C11H18NBF4
- Synonyms: HexPy BF4, N-Hexylpyridinium tetrafluoroborate
- Anion decomposes slowly in the presence of water.
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 0.90 mS/cm (30 °C)
SUM Formula: C11H18NBF4
Molecular Weight: 251.07
Melting Point: <RT
Density: 1.16 g/cm³ (23 °C)
Purity: >99%
Viscosity: 460 cP (18 °C)
- SUM Formula: C11H18NBF4
- Molecular Weight: 251.07
- Melting Point:
- Density: 1.16 g/cm³ (23 °C)
- Viscosity: 460 cP (18 °C)
1-Hexylpyridinium tetrafluoroborate, CAS: 474368-70-2
Key Applications:
Electrochemistry & Energy Systems
- Electrolyte component in supercapacitors and high‑voltage electrochemical cells where enhanced ionic conductivity and stability are required.
- Solvent/electrolyte medium for electrodeposition of metals and alloys, enabling smoother films and improved morphology control.
- Redox‑active media for studying charge‑transfer kinetics in non‑aqueous systems.
Catalysis & Organic Synthesis
- Reaction medium for transition‑metal‑catalyzed transformations, including C–C and C–N coupling, benefiting from the ionic liquid’s polarity and thermal robustness.
- Phase‑transfer environment that enhances reaction rates and selectivity in biphasic catalytic systems.
- Stabilizing medium for nanoparticle‑based catalysts, supporting dispersion and preventing aggregation.
Separation Science & Extraction
- Hydrophobic extraction solvent for selective removal of aromatic hydrocarbons, metal ions, and polar organics.
- Supported ionic liquid phase (SILP) component for gas separation membranes, improving permeability and selectivity.
- Liquid–liquid extraction of bioactive compounds and specialty chemicals where low vapor pressure and tunable polarity are advantageous.
Materials Science & Surface Engineering
- Template/structuring agent in the synthesis of porous materials, nanocomposites, and functional coatings.
- Additive in polymer formulations to modify conductivity, mechanical properties, and thermal behavior.
- Surface‑modification medium for creating ion‑rich interfaces on metals, ceramics, and carbon materials.
Analytical & Spectroscopic Applications
- Solvation medium for NMR, UV‑Vis, and electroanalytical studies requiring stable, non‑volatile ionic environments.
- Matrix component in studying solute–solvent interactions, ion pairing, and transport phenomena.
Contact us if you want to learn more or need assistance with your order.