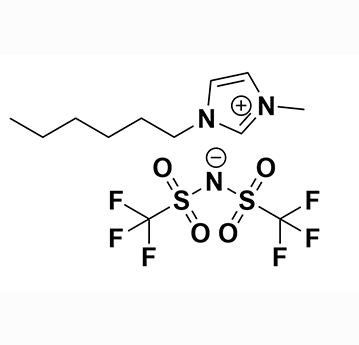
1-Hexyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, >99.5%
Price range: $190.97 through $2,604.47
Product Code: IL-0098-UPCAS NO: 382150-50-7
- Chemical Formula: C12H19F6N3O4S2
- Synonyms: HMIM BTA, HMIM NTf 2, HMIM TFSI, HMIM BTI, C 1 C 6 Im BTA, Im 16 BTA
- Weakly-coordinating anion.
- Hydrophobic
- Aromatic
For High Purity of >99%, refer to IL-0098-HP
Conductivity: 1.73 mS/cm (20 °C)
SUM Formula: C12H19F6N3O4S2
Molecular Weight: 447.42
Melting Point: -7 °C
Density: 1.37 g/cm³ (29 °C)
ECW: 5.3V
Purity: >99.5%
Viscosity: 88 cP (20 °C)
- SUM Formula: C12H19F6N3O4S2
- Molecular Weight: 447.42
- Melting Point: -7 °C
- Density: 1.37 g/cm³ (29 °C)
- ECW: 5.3V
- Viscosity: 88 cP (20 °C)
1-Hexyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 382150-50-7
Key Applications:
1. Gas Separation and CO₂ Capture
CO₂ Selective Solvent
- The long C6 alkyl chain increases free volume and enhances CO₂ solubility relative to shorter‑chain imidazolium ILs.
- The TFSI⁻ anion provides high fluorination, which strengthens quadrupole interactions with CO₂, improving absorption capacity.
- Suitable for physical absorption processes, membrane‑supported IL phases, and hybrid IL–polymer systems.
- Ultra‑high purity (>99.5%) ensures reproducible solubility data, critical for modeling and scale‑up in carbon capture R&D.
Supported Ionic Liquid Membranes (SILMs)
- High hydrophobicity and low vapor pressure minimize IL loss during long‑term CO₂/N₂ or CO₂/CH₄ separation.
- Stability under pressure differentials makes it attractive for pilot‑scale membrane modules.
2. Adsorption and Surface‑Mediated Processes
Surface Functionalization and Tunable Adsorption
- Acts as a surface modifier for porous carbons, silica, and metal–organic frameworks (MOFs), improving CO₂ affinity and selectivity.
- The C6 chain enhances dispersibility on hydrophobic surfaces, enabling uniform IL coatings.
- High purity reduces interference from trace halides or moisture, which can alter adsorption isotherms.
Ion‑Pair–Driven Adsorption Studies
- Used as a benchmark IL for studying ion–surface interactions, wettability, and adsorption kinetics in advanced materials research.
- Supports development of IL‑gated electrodes and IL‑modified sorbents.
3. Solvent and Reaction Medium
Non‑Volatile, Thermally Stable Solvent
- Ideal for moisture‑sensitive or high‑temperature reactions due to its wide electrochemical and thermal stability windows.
- Commonly used in organometallic catalysis, transition‑metal complex stabilization, and biphasic catalysis.
Solvent for Electrochemical and Spectroscopic Studies
- High ionic conductivity and low viscosity (relative to longer‑chain ILs) make it suitable for electrochemical measurements.
- Ultra‑high purity minimizes background signals in NMR, FTIR, and UV‑Vis studies.
4. Energy Storage and Electrolyte Systems
Lithium Battery Electrolytes
- TFSI⁻ anion provides excellent electrochemical stability and compatibility with Li salts.
- Used in Li‑ion, Li‑metal, and solid‑state battery research as a co‑solvent or primary electrolyte component.
- High purity reduces degradation pathways that shorten cycle life.
Supercapacitors and Ionic‑Liquid‑Based EDLCs
- High voltage stability supports wide electrochemical windows for high‑energy supercapacitors.
- Low volatility ensures safe operation under elevated temperatures.
5. Materials Processing and Nanotechnology
Nanoparticle Synthesis and Stabilization
- Serves as a templating medium for metal, metal oxide, and carbon nanostructures.
- Hydrophobicity and ion‑pair structure influence particle morphology and dispersion.
Polymer Processing
- Acts as a plasticizer or processing aid for fluoropolymers and high‑performance engineering plastics.
- Enhances ionic conductivity in polymer electrolytes.
6. Tribology and Lubrication
- Hydrophobic, thermally stable IL used as a boundary lubricant for steel, aluminum, and ceramic interfaces.
- Reduces wear and friction under high load due to strong surface adsorption of the imidazolium cation.
7. Analytical and Benchmark Uses
- High‑purity (>99.5%) grade is preferred for fundamental thermodynamic, viscosity, and conductivity measurements.
- Used as a reference IL in computational chemistry, MD simulations, and equation‑of‑state validation.
Please contact us if you want to learn more or need assistance with your order.