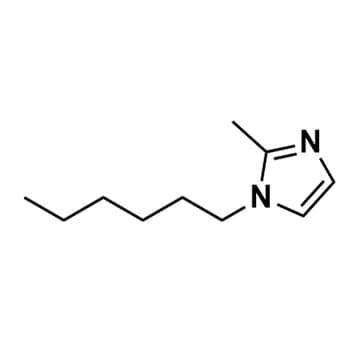
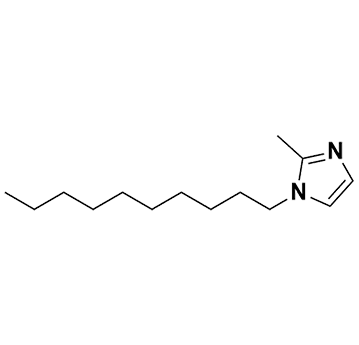
1-Hexyl-2-methylimidazole, >98%
Price range: $248.39 through $2,486.37
Product Code: KI-0011-HPCAS NO: 82410-68-2
- Chemical Formula: C10H18N2
- Synonyms: 1-Hex-2-MIM, 1-Hexyl-2-methyl-1 H-imidazole
- SMILES: CCCCCCN1C=CN=C1C.
SUM Formula: C10H18N2
Molecular Weight: 166.26
Purity: >98%
- SPECIFIC GRAVITY: NA
- SUM Formula: C10H18N2
- Molecular Weight: 166.26
- TSCA: NA
1-Hexyl-2-methylimidazole, CAS: 82410-68-2
Key Applications:
1. Precursor and Intermediate in Ionic Liquid Synthesis
- Serves as an alkylated imidazole building block for preparing hydrophobic ionic liquids with long‑chain cations.
- Useful for generating salts with bis(trifluoromethylsulfonyl)imide (TFSI), PF₆⁻, BF₄⁻, and other anions for electrochemical and materials applications.
- Enables tuning of viscosity, polarity, and thermal stability in task‑specific ionic liquids.
2. Corrosion Inhibition and Surface Protection
- Long alkyl chain improves adsorption on metal surfaces, making it a candidate for corrosion‑inhibitor formulations in acidic or chloride‑rich environments.
- Can be blended with other heterocyclic inhibitors to enhance film‑forming behavior.
3. Phase‑Transfer and Catalytic Applications
- Acts as a nucleophilic base or ligand in organic synthesis, particularly in alkylation, substitution, and condensation reactions.
- The hydrophobic C6 chain improves compatibility with nonpolar media, enabling biphasic or solvent‑free catalytic systems.
4. Surfactant and Amphiphilic Additive
- Amphiphilic structure supports micelle formation and interfacial modification.
- Can be incorporated into formulations requiring controlled wetting, dispersion, or stabilization.
5. Electrolyte and Additive in Energy‑Related Systems
- Serves as a precursor for cationic species used in electrolytes for batteries, supercapacitors, and electrochemical devices.
- The imidazole core contributes to ionic conductivity, while the hexyl chain enhances hydrophobicity and thermal robustness.
6. Polymer and Material Modification
- Functions as a reactive monomer or chain‑transfer component in specialty polymer systems.
- Can be grafted onto polymer backbones to introduce ionic or amphiphilic character, improving adhesion, flexibility, or chemical resistance.
7. Coordination and Organometallic Chemistry
- Nitrogen donor site enables coordination to transition metals, supporting catalyst development or mechanistic studies.
- Useful in designing metal–organic frameworks (MOFs) or functionalized ligands with tailored solubility.
Please Contact Us if you want to learn more or need assistance with your order.