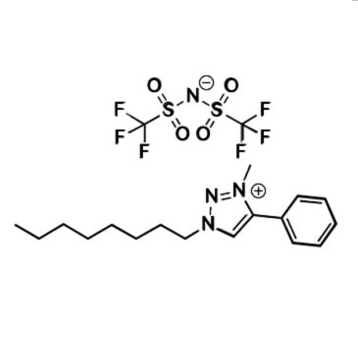
1-Hexyl-1,4-diaza[2.2.2]bicyclooctanium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $301.48 through $16,062.65
Product Code: IL-0314-HPCAS NO: 898256-50-3
- Chemical Formula: C16H23F9N2O3S
- Synonyms: HDABCO BTA, HDABCO NTf2, HDABCO TFSI, HDABCO BTI
- Weakly-coordinating anion
- Hydrophobic
SUM Formula: C16H23F9N2O3S
Molecular Weight: 477.49
Melting Point: 36 °C
Purity: >99%
- SUM Formula: C16H23F9N2O3S
- Molecular Weight: 477.49
- Melting Point: 36 °C
1-Hexyl-1,4-diaza[2.2.2]bicyclooctanium bis(trifluoromethylsulfonyl)imide, CAS: 898256-50-3
Key Application:
1. Electrolytes and Electrochemical Media
- Suitable for high‑voltage electrochemical systems due to the wide electrochemical window of TFSI⁻.
- Used in lithium, sodium, and multivalent battery research as an additive or co‑solvent to improve ionic conductivity and interfacial stability.
- Compatible with metal deposition studies, including controlled electrodeposition of transition metals.
- Serves as a stable medium for redox‑active species in flow‑battery research.
2. Ion Transport and Conductivity Modifiers
- The bicyclic diammonium core provides high cationic mobility, making it useful in designing ionic‑liquid‑based ionogels and polymer electrolytes.
- Enhances mechanical stability and ionic transport in solid‑state electrolyte matrices.
3. Phase‑Transfer and Separation Processes
- Effective in biphasic catalysis and extraction systems due to its hydrophobic TFSI⁻ anion.
- Useful for selective extraction of metal ions, particularly soft or highly charged species.
- Can function as a tunable solvent for CO₂ capture or gas‑separation studies.
4. Catalysis and Reaction Media
- Provides a non‑volatile, thermally stable medium for organic synthesis under high‑temperature or microwave conditions.
- Supports acid‑catalyzed and metal‑catalyzed transformations where conventional solvents degrade.
- The rigid bicyclic cation can influence reaction selectivity in task‑specific catalytic systems.
5. Materials Science and Surface Engineering
- Used in preparing thin films, coatings, and ionically conductive layers.
- Compatible with polymer blending, enabling antistatic, conductive, or ion‑exchange properties.
- Can act as a templating agent for nanoporous materials.
6. Analytical and Spectroscopic Applications
- Serves as a stable matrix for mass spectrometry ionization studies.
- Useful in electroanalytical experiments requiring low vapor pressure and high thermal stability.
Please contact us if you want to learn more or need assistance with your order.

![1-Hexyl-1,4-diaza[2.2.2]bicyclooctanium bis(trifluoromethylsulfonyl)imide,898256-50-3](https://mlhxrkjoggqr.i.optimole.com/w:359/h:345/q:mauto/f:best/https://roco.global/wp-content/uploads/2022/11/IL-0314-HP.png)