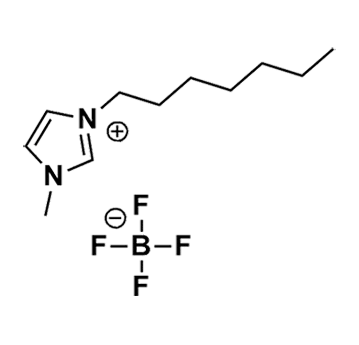
1-Heptyl-3-methylimidazolium tetrafluoroborate, >99%
Price range: $229.30 through $13,403.06
Product Code: IL-0304-HPCAS NO: 244193-51-9
- Chemical Formula: C11H21BF4N2
- Synonyms: HeptMIM BF4, C1C7Im BF4, Im17 BF4
- Anion decomposes slowly in the presence of water
- Hydrophobic
- Aromatic
SUM Formula: C11H21BF4N2
Molecular Weight: 268.10
Melting Point: <RT
Purity: >99%
- SUM Formula: C11H21BF4N2
- Molecular Weight: 268.10
- Melting Point:
1-Heptyl-3-methylimidazolium tetrafluoroborate, CAS: 244193-51-9
Key Applications:
Electrochemical and Energy‑Related Uses
- Electrolyte component in supercapacitors and electrochemical cells
The long heptyl chain improves viscosity–conductivity balance, while the BF₄⁻ anion supports wide electrochemical windows.
- Medium for electrodeposition of metals and alloys
Useful for smoother, more uniform films compared to aqueous systems.
- Solvent for redox‑active species in fundamental electrochemical studies.
Catalysis and Synthesis
- Reaction medium for acid‑catalyzed and transition‑metal‑catalyzed transformations
The ionic environment can enhance selectivity and stabilize reactive intermediates.
- Phase‑transfer–like behavior in biphasic systems
The heptyl chain increases organic compatibility, enabling improved mass transfer.
- Support for immobilized catalysts
Forms stable ionic‑liquid films on solid supports.
Extraction and Separation
- Liquid–liquid extraction of aromatic hydrocarbons, dyes, and metal ions
Hydrophobicity aids partitioning of nonpolar or moderately polar solutes.
- CO₂ and SO₂ absorption studies
BF₄⁻ systems are often used as benchmarks for gas‑solubility comparisons.
- Selective extraction of rare‑earth or transition‑metal ions when paired with coordinating ligands.
Materials and Surface Science
- Template or structuring agent in nanoparticle synthesis
Controls particle size and dispersion due to strong ionic microenvironments.
- Additive in polymer electrolytes or ion‑conducting films
Enhances ionic mobility and thermal stability.
- Surface modification medium for creating IL‑coated electrodes or membranes.
Spectroscopy and Fundamental Studies
- Solvent for NMR, IR, and Raman investigations of solvation dynamics
The heptyl chain provides tunable polarity and microheterogeneity.
- Model system for studying cation–anion interactions and nanostructuring in ionic liquids.
Please contact us if you want to learn more or need assistance with your order.