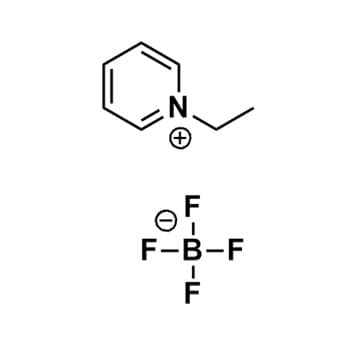
1-Ethylpyridinium tetrafluoroborate, >99%
Price range: $355.25 through $3,530.05
Product Code: IL-0107-HPCAS NO: 350-48-1
- Chemical Fr: C7H10BF4N
- Synonyms: EtPy BF4, N-Ethylpyridinium tetrafluoroborate
- Anion decomposes slowly in the presence of water
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
SUM Formula: C7H10BF4N
Molecular Weight: 194.97
Melting Point: 46 °C
Purity: >99%
- SPECIFIC GRAVITY: NA
- SUM Formula: C7H10BF4N
- Molecular Weight: 194.97
- Melting Point: 46 °C
1-Ethylpyridinium tetrafluoroborate, CAS: 350-48-1
Key Applications:
Electrochemical Systems
- Electrolyte component in supercapacitors and electrochemical double‑layer capacitors (EDLCs), where its ionic mobility and wide liquid‑phase range enhance charge–discharge stability.
- Supporting electrolyte for electrodeposition of metals (e.g., Cu, Ni, Ag) under water‑sensitive or morphology‑controlled conditions.
- Medium for redox‑active species, improving solubility and diffusion in electrochemical sensors and analytical devices.
Catalysis & Organic Synthesis
- Reaction medium for acid‑catalyzed, transition‑metal‑catalyzed, and biphasic transformations requiring low volatility and thermal stability.
- Solvent for nucleophilic substitution, alkylation, and cyclization reactions, enabling improved yields and simplified product isolation.
- Stabilizing environment for homogeneous catalysts, particularly in systems where BF₄⁻ supports weakly coordinating conditions.
Materials Science & Nanostructures
- Template and stabilizing medium for nanoparticle synthesis (e.g., Au, Pt, metal oxides), enabling controlled particle size and dispersion.
- Processing aid in polymer electrolytes and ion‑conducting films, improving ionic transport and mechanical stability.
- Additive in sol–gel and thin‑film fabrication, where its ionic character influences porosity and microstructure.
Separation Science
- Extractant or co‑solvent in liquid–liquid extraction of aromatic hydrocarbons, metal ions, and polar organics.
- Stationary‑phase modifier in chromatography, enhancing selectivity for polar and aromatic analytes.
- Medium for CO₂ and SO₂ capture studies, leveraging pyridinium cation interactions with acidic gases.
Thermal & Green Chemistry Applications
- Heat‑transfer and thermal‑storage fluid in moderate‑temperature systems due to its low vapor pressure and thermal robustness.
- Replacement for volatile organic solvents in sustainable processing, reducing emissions and improving operator safety.
Contact us if you want to learn more or need assistance with your order.