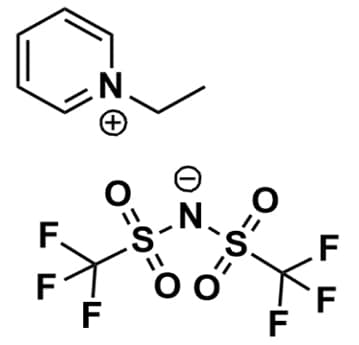
1-Ethylpyridinium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $220.76 through $13,455.34
Product Code: IL-0211-HPCAS NO: 244193-65-5
- Chemical Formula: C9 H10 F6 N2 O4 S2
- Synonyms: N-Ethylpyridinium bis(trifluoromethylsulfonyl)imide, EtPy BTA, EtPy NTf2, EtPy TFSI, EtPy BTI
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 5.545 mS/cm
SUM Formula: C9 H10 F6 N2 O4 S2
Molecular Weight: 388.31
Melting Point: 32 °C
Density: 1.586 g/cm³
Purity: >99%
Viscosity: 40.5 cP
- SUM Formula: C9 H10 F6 N2 O4 S2
- Molecular Weight: 388.31
- Melting Point: 32 °C
- Density: 1.586 g/cm³
- Viscosity: 40.5 cP
1-Ethylpyridinium bis(trifluoromethylsulfonyl)imide, CAS: 244193-65-5
Key Applications:
Electrolytes for Energy Storage
• High‑conductivity ionic liquid electrolyte for lithium‑ion, sodium‑ion, and magnesium‑ion batteries, especially in high‑temperature or high‑voltage environments.
• Component in supercapacitor electrolytes, improving thermal stability and widening the electrochemical window.
• Used in redox‑flow battery research where low volatility and strong anion stability are required.
Catalysis & Organic Synthesis
• Polar, thermally stable reaction medium for Friedel–Crafts, nucleophilic substitutions, and transition‑metal‑catalyzed transformations.
• Enhances selectivity in biphasic catalytic systems, enabling efficient product separation.
• Employed as a non‑coordinating ionic environment for organometallic catalysis and mechanistic studies.
High‑Temperature & High‑Stability Solvent
• Ideal for thermally demanding reactions due to its low vapor pressure and exceptional oxidative stability.
• Used in spectroscopic studies (NMR, IR, electrochemical) requiring inert, non‑volatile ionic media.
Electrochemical & Surface Science
• Electrolyte for electrodeposition of metals and alloys, particularly where moisture‑free conditions are essential.
• Medium for electrochemical impedance spectroscopy (EIS) and cyclic voltammetry in fundamental ion‑transport studies.
• Supports formation of stable electric double layers in advanced electrochemical interfaces.
Separation, Extraction & Purification
• Tunable solvent for liquid–liquid extraction of metal ions, dyes, and organic contaminants.
• Applied in CO₂ capture, SO₂ absorption, and gas‑separation research due to the TFSI⁻ anion’s hydrophobicity.
• Used in ionic‑liquid‑based chromatography as a stationary‑phase modifier.
Polymer & Materials Engineering
• Additive or processing aid in ion‑conducting polymers, polymer electrolytes, and block‑copolymer self‑assembly.
• Plasticizer for high‑performance elastomers and specialty materials requiring thermal stability.
• Employed in nanomaterials synthesis, including templated nanoparticle growth and surface functionalization.
Fundamental Research
• Model ionic liquid for studying cation–anion interactions, solvation dynamics, and ion transport.
• Benchmark system in molecular dynamics simulations and physicochemical property mapping (viscosity, conductivity, density).
Please contact us if you want to learn more or need assistance with your order.