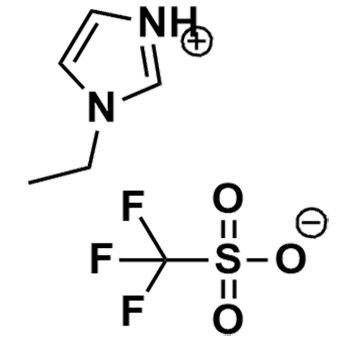
1-Ethylimidazolium triflate, >98%
Price range: $280.22 through $2,220.75
Product Code: IL-0271-HPCAS NO: 501693-46-5
- Chemical Formula: C6H9F3N2O3S
- Synonyms: EIM OTf, 1-Ethylimidazolium trifluoromethanesulfonate
- Hydrophobic
- Aromatic
- Protic
**This product will incur a $97.00 HazMat fee when the order is placed.**
Conductivity: 0.81 ms/cm (27 °C)
SUM Formula: C6H9F3N2O3S
Molecular Weight: 246.21
Melting Point: 27 °C
Density: 1.355 g/cm³ (26 °C)
Purity: >98%
Viscosity: 78 cP (30 °C)
- SUM Formula: C6H9F3N2O3S
- Molecular Weight: 246.21
- Melting Point: 27 °C
- Density: 1.355 g/cm³ (26 °C)
- Viscosity: 78 cP (30 °C)
1-Ethylimidazolium triflate, CAS: 501693-46-5
Key Applications:
Catalysis and Organic Synthesis
- Serves as a non‑volatile, thermally stable medium for acid‑catalyzed reactions.
- Supports esterification, alkylation, and dehydration processes where a strong, non‑nucleophilic anion is advantageous.
- Useful as a solvent or co‑solvent for transition‑metal catalysis due to its high ionic conductivity and low coordinating behavior.
Electrochemistry and Energy Materials
- Functions as an electrolyte component in electrochemical cells, particularly where wide electrochemical windows and low vapor pressure are required.
- Enhances ion transport in redox systems and can be blended with other ionic liquids to tune viscosity and conductivity.
Materials Science and Polymer Processing
- Acts as a processing aid for polymer dissolution, modification, and ion‑exchange functionalization.
- Enables controlled incorporation of ionic domains into polymer matrices for membranes, ionogels, and specialty coatings.
Separation Science
- Supports selective extraction of metal ions, organic compounds, and catalytic species due to its tunable polarity and thermal stability.
- Useful in biphasic systems where immiscibility with nonpolar solvents facilitates product recovery.
Spectroscopy and Analytical Chemistry
- Provides a stable, low‑volatility medium for NMR, IR, and electroanalytical studies.
- Reduces background interference in systems requiring non‑coordinating anions.
Green Chemistry and Process Intensification
- Replaces volatile organic solvents in high‑temperature or moisture‑sensitive reactions.
- Enables solvent recycling and reduced emissions in closed‑loop processes.
Please contact us if you want to learn more or need assistance with your order.