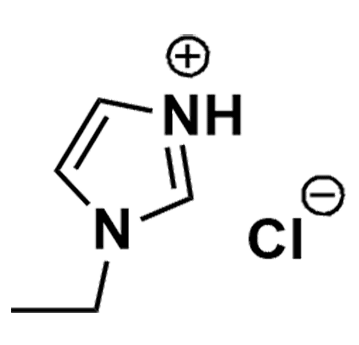
1-Ethylimidazolium chloride, >98%
Price range: $91.96 through $705.59
Product Code: IL-0275-SGCAS NO: 81505-35-3
- Chemical Formula: C5H9ClN2
- Synonyms: EIM Cl, EtIm Cl
- Hydrophilic
- Aromatic
- Protic
To order 5kg or more, please email us at info@roco.global
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C5H9ClN2
Molecular Weight: 132.59
Melting Point: 67 °C
Density: 1.223 g/cm³ (26 °C)
Purity: >98%
- SUM Formula: C5H9ClN2
- Molecular Weight: 132.59
- Melting Point: 67 °C
- Density: 1.223 g/cm³ (26 °C)
1-Ethylimidazolium chloride, CAS 81505-35-3
Key Applications:
Hydrogen‑bonding and solvation behavior
- The imidazolium N–H group functions as a hydrogen‑bond donor, while chloride is a strong hydrogen‑bond acceptor.
- The resulting directional hydrogen‑bond network enhances solvation of polar, protic, and hydrogen‑bonding solutes.
- High polarity and strong association support catalytic and extraction systems requiring robust solute–solvent interactions.
Catalysis and acid‑base chemistry
- Effective medium for Brønsted‑acid catalysis due to proton‑transfer capability and chloride stabilization of intermediates.
- Supports nucleophilic substitution, dehydration, esterification, and biomass‑related transformations where chloride activates substrates or stabilizes transition states.
- Hydrogen‑bonding environment can accelerate reactions involving polar or charged intermediates.
Biomass processing and dissolution
- Strong hydrogen‑bonding capacity disrupts cellulose and lignocellulosic hydrogen‑bond networks.
- Enables swelling, partial dissolution, or pretreatment of biomass to improve downstream enzymatic or chemical conversion.
- Chloride‑based ionic liquids are among the most effective for reducing cellulose crystallinity.
Extraction and separation
- Suitable for extracting metal ions, polar organics, and hydrogen‑bonding solutes through chloride coordination or hydrogen bonding.
- Applicable in aqueous biphasic systems where hydrogen bonding governs phase behavior and selectivity.
- Useful for separating alcohols, amines, and other protic compounds.
Electrochemical and materials applications
- High‑polarity electrolyte component for systems requiring strong ion–solvent interactions.
- Hydrogen bonding influences ion mobility and conductivity, relevant for protic ionic liquid electrolytes, electrodeposition baths, and polymer electrolyte formulations.
- Can act as a structure‑directing medium in materials synthesis.
Solvent for spectroscopy and mechanistic studies
- Directional hydrogen‑bond network makes it a model system for studying solvation dynamics, proton transfer, and ion pairing.
- Stabilizes reactive intermediates or charged species in NMR, IR, and kinetic studies.
Please contact us if you want to learn more or need assistance with your order.