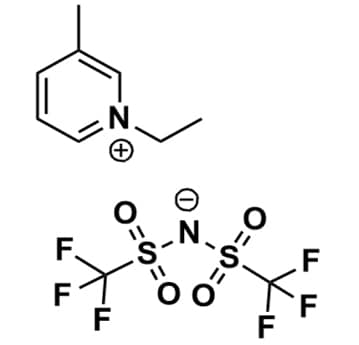
1-Ethyl-3-methylpyridinium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $149.93 through $8,208.21
Product Code: IL-0214-HPCAS NO: 841251-37-4
- Chemical Formula: C10H12F6N2O4S2
- Synonyms: Et-3-Pic BTA, Et-3-Pic NTf2, Et-3-Pic TFSI, Et-3-Pic BTI, Et3Pic BTA, Et3Pic NTf2, Et3Pic TFSI, Et3Pic BTI, Ethyl-3-picolinium bis(trifluoromethylsulfonyl)imide, N-Ethyl-3-methylpyridinium bis(trifluoromethylsulfonyl)imide, N-Ethyl-3-picolinium bis(trifluoromethylsulfonyl)imide
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 7.47 mS/cm (30 °C)
SUM Formula: C10H12F6N2O4S2
Molecular Weight: 402.33
Melting Point: 6 °C
Density: 1.48 g/cm³ (26 °C)
Purity: >99%
Viscosity: 36.6 cP (25 °C)
- SUM Formula: C10H12F6N2O4S2
- Molecular Weight: 402.33
- Melting Point: 6 °C
- Density: 1.48 g/cm³ (26 °C)
- Viscosity: 36.6 cP (25 °C)
1-Ethyl-3-methylpyridinium bis(trifluoromethylsulfonyl)imide, CAS: 841251-37-4
Key Applications:
Electrochemical Systems
- Serves as a high‑stability ionic liquid electrolyte for lithium, sodium, and multivalent metal batteries.
- Provides wide electrochemical windows and low volatility, supporting high‑voltage cell designs.
- Used as a conductive medium in supercapacitors, enabling improved ionic mobility and thermal resilience.
- Functions as a stable solvent/electrolyte for electrodeposition of metals and alloys under moisture‑sensitive or high‑temperature conditions.
Catalysis and Organic Synthesis
- Acts as a non‑volatile, thermally robust reaction medium for transition‑metal‑catalyzed transformations.
- Supports acid‑sensitive and base‑sensitive reactions due to its weakly coordinating TFSI anion.
- Enables biphasic catalysis and catalyst recycling workflows.
- Useful for selective alkylation, nucleophilic substitution, and oxidative coupling reactions requiring controlled ionic environments.
Separation Science and Extraction
- Employed in liquid–liquid extraction of metal ions, particularly rare‑earth and actinide species, due to its hydrophobicity and tunable coordination behavior.
- Used in chromatographic method development as an ionic liquid additive to improve peak shape and separation efficiency.
- Supports gas absorption studies, including CO₂ solubility and transport behavior in fluorinated ionic liquids.
Materials Science and Polymer Engineering
- Incorporated into polymer matrices to enhance ionic conductivity, flexibility, and thermal stability.
- Used in ionogel formation for flexible electronics, sensors, and solid‑state electrochemical devices.
- Serves as a plasticizer or mobility‑enhancing additive in high‑performance polymer electrolytes.
Surface Science and Coatings
- Provides controlled interfacial behavior for electrode modification, corrosion studies, and surface passivation research.
- Used as a medium for forming uniform, defect‑minimized thin films via electrochemical or solution‑based deposition.
- Supports lubrication studies where low vapor pressure and high thermal stability are required.
Spectroscopy and Fundamental Research
- Employed as a reference ionic liquid for benchmarking transport properties (viscosity, conductivity, diffusion).
- Used in vibrational, NMR, and dielectric spectroscopy to study ion pairing, solvation, and dynamic relaxation processes.
- Supports computational and experimental investigations of structure–property relationships in pyridinium‑based ionic liquids.
Please contact us if you want to learn more or need assistance with your order.