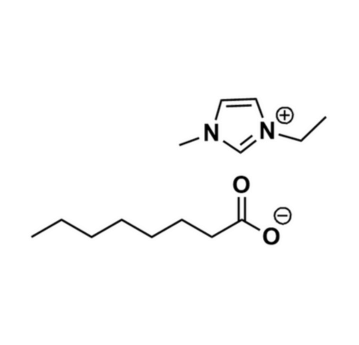
1-Ethyl-3-methylimidazolium octanoate, >98%
Price range: $41.04 through $672.31
Product Code: IL-0357-HPCAS NO: 1154003-55-0
- Chemical Formula: C14H26N2O2
- Synonyms: EMIM Oct, EMIM OOc, EMIM Oc, C1C2Im Oct
Conductivity: 0.20 mS/cm (19 °C)
SUM Formula: C14H26N2O2
Molecular Weight: 254.38
Melting Point: -30 °C
Density: 1.040 g/cm³ (19 °C)
ECW: 5.0 V
Purity: >98%
Viscosity: 799.1 cP (19 °C)
- SUM Formula: C14H26N2O2
- Molecular Weight: 254.38
- Melting Point: -30 °C
- Density: 1.040 g/cm³ (19 °C)
- ECW: 5.0 V
- Viscosity: 799.1 cP (19 °C)
1-Ethyl-3-methylimidazolium octanoate, CAS: 1154003-55-0
Key Applications:
Biomass Processing and Cellulose Solvation
- Effective cellulose solvent due to the combined hydrogen‑bond‑disrupting capability of the imidazolium cation and the moderately hydrophobic octanoate anion.
- Supports dissolution, swelling, and regeneration of cellulose, enabling film formation, fiber spinning, and derivatization workflows.
- Useful in lignocellulosic pretreatment, selectively solubilizing lignin and hemicellulose to improve downstream enzymatic hydrolysis.
Catalysis and Reaction Media
- Provides a medium‑polarity ionic environment suitable for base‑catalyzed, organometallic, and condensation reactions.
- Enhances phase‑transfer behavior in biphasic systems due to the amphiphilic nature of the octanoate anion.
- Stabilizes reactive intermediates in esterification, transesterification, and related transformations.
Surfactant, Emulsion, and Formulation Science
- Exhibits intrinsic amphiphilicity, enabling formation of microemulsions and stabilization of dispersions.
- Functions as a co‑solvent or co‑surfactant in formulations requiring tunable polarity and low volatility.
- Improves nanoparticle dispersion for metal oxides, carbon nanomaterials, and hybrid composites.
Electrochemical and Energy Applications
- Offers moderate hydrophobicity with useful ionic conductivity for low‑voltage electrochemical systems.
- Serves as an ion‑transport medium in polymer electrolytes, gels, and flexible electronic materials.
- Suitable for bioelectrochemical devices where carboxylate anions provide lower cytotoxicity than halides.
Biocompatible and Bio‑derived Systems
- Carboxylate anions such as octanoate are generally less cytotoxic, enabling use in enzyme stabilization, protein extraction, and selective microbial phase systems.
- Supports bioprocessing environments where ionic liquids enhance solubility without denaturing biomolecules.
Polymer and Materials Processing
- Acts as a plasticizer or processing aid for biopolymers including cellulose, chitosan, and PLA blends.
- Enables formation of ionically conductive polymer films for sensors and soft electronics.
- Useful in surface modification workflows where hydrophobic chain length improves adsorption and interfacial behavior.
Please contact us if you want to learn more or need assistance with your order.