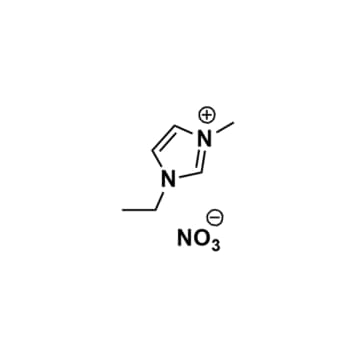
1-Ethyl-3-methylimidazolium nitrate, >98%
Price range: $284.55 through $4,548.70
Product Code: IL-0005-HPCAS NO: 143314-14-1
- Chemical Formula: C6H11N3O3
- Synonyms: EMIM NO3, C1C2Im NO3, Im12 NO3
- Hydrophilic
- Aromatic
*This product will incur a $97.00 HazMat fee when the order is placed.*
SUM Formula: C6H11N3O3
Molecular Weight: 173.17
Melting Point: 38 °C
Density: 1.25 g/cm³
Purity: >98%
Viscosity: 21.3 cP (45 °C)
- SUM Formula: C6H11N3O3
- Molecular Weight: 173.17
- Melting Point: 38 °C
- Density: 1.25 g/cm³
- Viscosity: 21.3 cP (45 °C)
1-Ethyl-3-methylimidazolium nitrate, CAS: 143314-14-1
Key Applications:
1. Biomass Processing and Lignocellulose Dissolution
- Disrupts hydrogen‑bond networks in cellulose and lignin.
- Used in pretreatment workflows for biofuel, biopolymer, and biorefinery research.
- Supports homogeneous reaction environments for derivatization processes such as acetylation, etherification, and grafting.
2. Electrochemical Systems
- Functions as a non‑volatile ionic medium for redox chemistry and electrodeposition studies.
- Provides stable ionic conductivity in moderate‑temperature electrochemical cells.
- Investigated as a component in nitrate‑based electrolytes for metal‑ion batteries and supercapacitors.
3. Catalysis and Reaction Media
- Serves as a polar, thermally stable solvent for transition‑metal catalysis.
- Enhances selectivity in oxidation, nitration, and coupling reactions due to the nitrate anion.
- Supports biphasic catalysis and catalyst recycling strategies.
4. Heat‑Transfer and Thermal‑Stability Research
- High thermal stability and negligible vapor pressure make it suitable for heat‑transfer fluid studies.
- Used in modeling ionic‑liquid thermophysical properties including viscosity, conductivity, and heat capacity.
5. Green Chemistry and Solvent Replacement
- Replaces volatile organic solvents in extraction, dissolution, and synthesis workflows.
- Compatible with microwave‑assisted reactions and energy‑efficient processing.
6. Materials Science and Nanomaterials
- Acts as a templating or stabilizing medium for nanoparticle synthesis, including metal and metal‑oxide systems.
- Supports controlled nucleation and growth due to its ionic environment.
- Used in polymer electrolyte and ionogel fabrication.
7. Environmental and Separation Technologies
- Applied in selective extraction of metal ions, dyes, and organic pollutants.
- Useful in liquid–liquid extraction systems where nitrate coordination enhances selectivity.
Please contact us if you want to learn more or need assistance with your order.
Additional information
| Weight | N/A |
|---|---|
| Qty | 25 Grams, 50 Grams, 100 Grams, 250 Grams, 500 Grams, 1000 Grams, 5000 Grams |