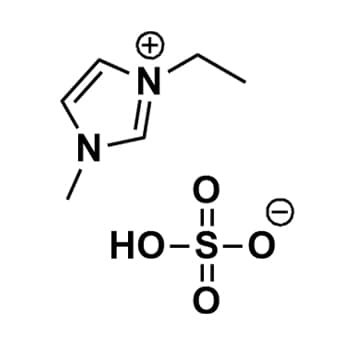
1-Ethyl-3-methylimidazolium hydrogensulfate, >98%
Price range: $127.31 through $824.95
Product Code: IL-0091-HPCAS NO: 412009-61-1
- Chemical Fr: C6H12N2O4S
- Synonyms: 1-Ethyl-3-methylimidazolium bisulfate, EMIM HSO4, C1C2Im HSO4, Im12 HSO4
- Aromatic
To order 5kg or more, please email us at info@roco.global
** This product will incur a $97.00 HazMat fee when the order is placed. **
Conductivity: 0.50 mS/cm (20 °C):
SUM Formula: C6H12N2O4S
Molecular Weight: 208.24
Melting Point: -23 °C
Density: 1.36 g/cm³ (28 °C)
Purity: >98%
Viscosity: 1510 cP (25 °C)
- SUM Formula: C6H12N2O4S
- Molecular Weight: 208.24
- Melting Point: -23 °C
- Density: 1.36 g/cm³ (28 °C)
- Viscosity: 1510 cP (25 °C)
1-Ethyl-3-methylimidazolium hydrogensulfate, CAS: 412009-61-1
Key Applications:
Catalysis and Acid‑Driven Transformations
- Functions as a Brønsted acidic ionic liquid, enabling esterification, transesterification, alkylation, and dehydration reactions.
- Strong hydrogen‑bonding capacity stabilizes transition states and enhances reaction rates in both neat and aqueous media.
- Serves as a recyclable, low‑volatility alternative to mineral acids in homogeneous catalysis.
Biomass Processing and Fractionation
- Effective for cellulose swelling, partial dissolution, and hydrolysis, supported by its hydrogen‑bonding interactions with polysaccharide networks.
- Facilitates lignocellulosic pretreatment in aqueous or mixed aqueous–ionic liquid systems, reducing viscosity and improving mass transfer.
- Supports acid‑catalyzed conversion of biomass‑derived intermediates (e.g., furans, levulinates).
Electrochemical and Energy Applications
- Used as a proton‑conducting medium in electrochemical devices due to its intrinsic acidity and hydrogen‑bond network.
- Enhances ionic conductivity in aqueous electrolyte blends, improving stability and widening the operational temperature window.
- Suitable for electrodeposition and corrosion‑inhibition studies where acidic ionic environments are required.
Separation Science and Extraction
- Applied in liquid–liquid extraction of metal ions, dyes, and organics, with hydrogen bonding enabling selective solvation.
- Performs well in aqueous biphasic systems, where tunable acidity and hydrophilicity support phase formation and targeted partitioning.
- Useful in desulfurization and purification workflows where proton activity aids contaminant removal.
Materials Processing and Synthesis
- Acts as a structure‑directing agent in the synthesis of inorganic and hybrid materials, leveraging its ionic and hydrogen‑bonding interactions.
- Supports polymer modification, protonation reactions, and acid‑assisted crosslinking in aqueous formulations.
- Provides a controlled acidic environment for nanoparticle synthesis and surface functionalization.
Green Chemistry and Process Engineering
- Replaces volatile acids in aqueous reaction systems, reducing corrosivity and improving operational safety.
- Demonstrates high thermal and chemical stability, enabling use in continuous‑flow or high‑temperature aqueous processes.
- Readily recyclable due to low vapor pressure and strong ionic cohesion.
Please contact us if you want to learn more or need assistance with your order.