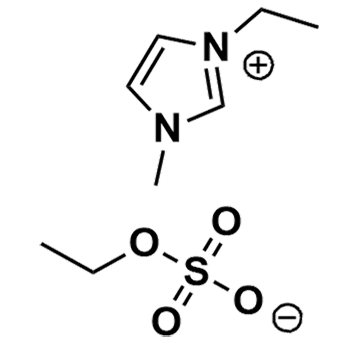
1-Ethyl-3-methylimidazolium ethyl sulfate, >98%
Price range: $59.46 through $3,406.94
Product Code: IL-0033-HPCAS NO: 342573-75-5
- Chemical Formula: C8H16N2O4S
- Synonyms: EMIM EtSO4 , C1 C2 Im EtSO4 , Im 12 EtSO 4
- Aromatic
- Hydrophilic
Conductivity: 2.97 mS/cm (25 °C)
SUM Formula: C8H16N2O4S
Molecular Weight: 236.29
Melting Point: -41 °C
Density: 1.24 g/cm³
ECW: 4.0V
Purity: >98%
Viscosity: 94.2 cP
- SUM Formula: C8H16N2O4S
- Molecular Weight: 236.29
- Melting Point: -41 °C
- Density: 1.24 g/cm³
- ECW: 4.0V
- Viscosity: 94.2 cP
1-Ethyl-3-methylimidazolium ethyl sulfate, CAS: 342573-75-5
Key Applications:
1. Biomass Processing & Green Chemistry
- Cellulose dissolution and pretreatment for biofuels, nanocellulose, and biopolymer production.
- Lignin fractionation and depolymerization, enabling selective extraction of aromatic monomers.
- Enhanced enzymatic hydrolysis due to reduced biomass recalcitrance after IL pretreatment.
- Solvent for carbohydrate transformations, including dehydration, esterification, and selective oxidation.
2. Acid‑Catalyzed Organic Transformations
- Esterification and transesterification (biodiesel, specialty esters) with improved reaction rates.
- Friedel–Crafts alkylation/acylation in low‑volatility, recyclable media.
- Dehydration reactions (e.g., alcohol → olefin) where the ethyl sulfate anion provides mild Brønsted acidity.
- Green solvent for multi‑step synthesis, replacing volatile organic solvents in bench‑scale and industrial processes.
3. Electrochemical & Energy Applications
- Electrolyte or co‑solvent in redox‑flow batteries, dye‑sensitized solar cells, and metal‑ion batteries.
- Stable ionic medium for electrodeposition of metals and alloys.
- Conductive additive in polymer electrolytes and ionogels, improving ionic transport and mechanical stability.
4. Catalysis & Reaction Engineering
- Task‑specific catalytic medium for acid‑driven reactions due to the sulfate anion’s proton‑donating character.
- Phase‑transfer catalyst substitute, improving mass transfer in biphasic systems.
- Support for immobilized catalysts, including enzymes and metal complexes.
5. Separation Science & Extraction
- Selective extraction of metal ions, especially transition metals and lanthanides.
- Solvent for liquid–liquid extraction of organic acids, phenolics, and dyes.
- Medium for CO₂ capture, leveraging strong solvation of acidic gases.
6. Formulations, Surfactants & Materials
- Additive in surfactant systems, improving micelle stability and solubilization of hydrophobic species.
- Plasticizer or processing aid for polymers, elastomers, and biopolymers.
- Stabilizing agent in nanoparticle synthesis (metal, metal oxide, carbon‑based).
- Ionogel precursor for flexible electronics, sensors, and membranes.
7. Thermal & Process Engineering
- Heat‑transfer fluid component due to high thermal stability and negligible vapor pressure.
- Solvent for high‑temperature reactions where conventional organics degrade.
- Medium for microwave‑assisted synthesis, benefiting from strong dielectric absorption.
Please contact us if you want to learn more or need assistance with your order.