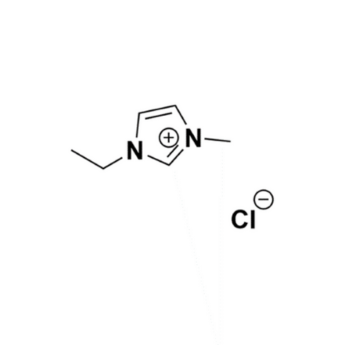
1-Ethyl-3-methylimidazolium chloride, >99%
Price range: $189.61 through $9,963.48
Product Code: IL-0093-UPCAS NO: 65039-09-0
- Chemical Formula: C6H11ClN2
- Synonyms: EMIM Cl, C1C2In Cl,In 12 Cl
- Aromatic
- Hydrophilic
For High-purity of >98%, refer to IL-0093-HP
For Purity of >95%, refer to IL-0093-TG
SUM Formula: C6H11ClN2
Molecular Weight: 146.62
Melting Point: 87°C
Density: 1.341 g/cm³ (26 °C)
Purity: >99%
- SUM Formula: C6H11ClN2
- Molecular Weight: 146.62
- Melting Point: 87°C
- Density: 1.341 g/cm³ (26 °C)
1-Ethyl-3-methylimidazolium chloride, CAS: 65039-09-0
Key Applications:
1. Biomass Dissolution and Processing
- Efficient solvent for cellulose, hemicellulose, and lignin dissolution.
- Enables homogeneous derivatization of cellulose (acetylation, etherification).
- Used in pretreatment steps for biofuel and biopolymer production.
2. Catalysis and Organic Synthesis
- Serves as a reaction medium for acid‑catalyzed and metal‑catalyzed transformations.
- Enhances selectivity in alkylation, esterification, and nucleophilic substitution reactions.
- Stabilizes reactive intermediates due to its ionic environment.
3. Electrochemistry and Materials Development
- Employed as an electrolyte component in electrochemical devices.
- Useful in electrodeposition of metals and semiconductors.
- Acts as a template or solvent for synthesizing nanomaterials and metal–organic frameworks.
4. Polymer Science
- Solvent for chitin, chitosan, and other biopolymers.
- Supports homogeneous polymer functionalization and blending.
- Used in membrane fabrication and ion‑conductive polymer development.
5. Extraction and Separation
- Effective for selective extraction of metal ions, dyes, and organic compounds.
- Applied in liquid–liquid extraction systems for rare‑earth and transition‑metal separation.
6. CO₂ Capture and Gas Solubility Studies
- Demonstrates moderate CO₂ solubility and is used in gas absorption research.
- Serves as a benchmark ionic liquid for studying gas–liquid equilibria.
7. Precursor for Ionic Liquid Synthesis
- Common starting material for preparing EMIM‑based ionic liquids via anion exchange.
- Used in metathesis reactions to generate hydrophobic ILs such as EMIM NTf₂ or EMIM PF₆.
Please contact us if you want to learn more or need assistance with your order.
Additional information
| Weight | 25 g |
|---|