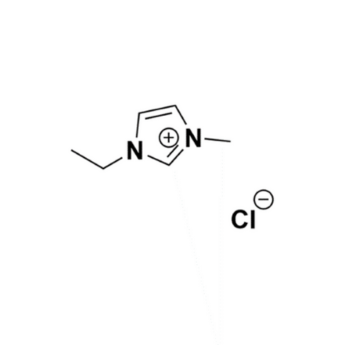
1-Ethyl-3-methylimidazolium chloride, >95%, melt
Price range: $70.70 through $2,167.00
Product Code: IL-0093-TGCAS NO: 65039-09-0
- Chemical Formula: C6H11ClN2
- Synonyms: EMIM Cl, C1C2Im Cl, Im12 Cl
For High purity of >98%, refer to IL-0093-HP
For Ultra-high purity of >99%, refer to IL-0093-UP
SUM Formula: C6H11ClN2
Molecular Weight: 146.62
Melting Point: 87 °C
Density: 1.341 g/cm³ (26 °C)
Purity: >95%
- SUM Formula: C6H11ClN2
- Molecular Weight: 146.62
- Melting Point: 87 °C
- Density: 1.341 g/cm³ (26 °C)
1-Ethyl-3-methylimidazolium chloride, CAS: 65039-09-0
Key Applications:
Deep Eutectic Solvents (DES) and Solvent Engineering
- Serves as a hydrogen‑bond acceptor for forming Type III DES with urea, glycerol, ethylene glycol, and other polyols.
- Enables tunable polarity and viscosity profiles for extraction, catalysis, biomass fractionation, and metal dissolution.
- Used in DES‑based pretreatment of lignocellulosic biomass to enhance delignification and sugar release.
- Supports metal‑ion complexation and selective dissolution in hydrometallurgical and recycling workflows.
SO₂ Absorption and Gas Handling
- Demonstrates high SO₂ solubility due to strong Lewis acidity and chloride coordination, making it suitable for reversible SO₂ capture.
- Applied in flue‑gas desulfurization research as a benchmark ionic liquid for absorption capacity and regeneration studies.
- Useful in mechanistic studies of acid‑gas interactions, including speciation, binding energetics, and absorption kinetics.
Conductivity and Electrochemical Systems
- Exhibits relatively high ionic conductivity among chloride‑based imidazolium salts, supporting use in electrochemical devices.
- Functions as a component in molten‑salt electrolytes for metal electrodeposition (Al, Zn, rare‑earth metals).
- Employed in ionic‑liquid‑based capacitors, redox‑flow systems, and conductivity‑tuning blends with other ILs or DES formulations.
- Used as a model chloride ionic liquid for studying ion transport, viscosity–conductivity relationships, and temperature‑dependent mobility.
Catalysis and Reaction Media
- Provides a polar, non‑volatile medium for acid‑catalyzed transformations, alkylation, esterification, and nucleophilic substitution.
- Stabilizes reactive intermediates and supports phase‑transfer catalysis in chloride‑rich environments.
- Enables metal‑catalyzed coupling reactions where chloride coordination influences catalyst speciation.
Materials Processing and Polymer Chemistry
- Acts as a solvent and swelling agent for cellulose, chitin, and other biopolymers.
- Used in polymer electrolyte formulations and ion‑conductive films.
- Supports nanoparticle synthesis where chloride availability influences nucleation and growth.
Metal Processing and Electroplating
- Provides a molten‑salt medium for electrodeposition of aluminum and other metals at moderate temperatures.
- Facilitates dissolution and separation of metal oxides and salts in recycling and refining workflows.
Analytical and Spectroscopic Uses
- Serves as a calibration and reference ionic liquid for conductivity, viscosity, and ion‑transport studies.
- Used in spectroscopic investigations of hydrogen bonding, ion pairing, and chloride coordination.
Please contact us if you want to learn more or need assistance with your order.