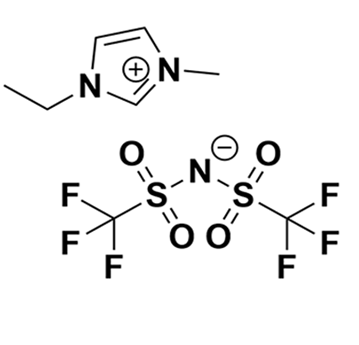
1-Ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, >99.5%
Price range: $77.88 through $1,109.64
Product Code: IL-0023-UPCAS NO: 174899-82-2
- Chemical Formula: C8H11F6N3O4S2
- Synonyms: EMIM BTA, EMIM NTf2, EMIM TFSI, EMIM BTI, C1C2Im BTA, Im12 BTA
- Weakly-coordinating anion.
- Hydrophobic
- Aromatic
For Optical Purity, colorless product, discover IL-0023-OP
**This product will incur a $97.00 HazMat fee when the order is placed.*
Conductivity: 6.63 mS/cm (20 °C)
SUM Formula: C8H11F6N3O4S2
Molecular Weight: 391.31
Melting Point: -17 °C
Density: 1.52 g/cm³
ECW: 4.7 V
Purity: >99.5%
Viscosity: 39.4 cP
- SUM Formula: C8H11F6N3O4S2
- Molecular Weight: 391.31
- Melting Point: -17 °C
- Density: 1.52 g/cm³
- ECW: 4.7 V
- Viscosity: 39.4 cP
1-Ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 174899-82-2
Key Applications:
Energy Storage: Batteries, Supercapacitors & Fuel Cells
- High‑stability electrolyte component for Li‑ion, Na‑ion, Mg‑ion, dual‑ion, and high‑voltage batteries
- Ionic liquid medium for supercapacitors (supercaps) requiring wide potential windows and low volatility
- Co‑solvent or additive in proton‑exchange and solid‑oxide fuel cells, improving ionic conductivity and durability
- Supports metal electrodeposition and interfacial‑kinetics studies in advanced battery chemistries
Dye‑Sensitized Solar Cells (DSSCs) & Optoelectronics
- Widely used electrolyte component in DSSCs, enabling stable I⁻/I₃⁻ redox shuttles
- Enhances charge transport, dye regeneration, and long‑term device stability
- Useful in tuning diffusion coefficients, viscosity, and interfacial recombination behavior
- Supports development of electrochromic devices and ion‑conducting optoelectronic films
Electrochemistry & Charge‑Transport Studies
- Benchmark ionic liquid for diffusion, ion mobility, and transport‑property measurements
- Solvent for redox chemistry, electrosynthesis, and electrode‑surface studies
- Medium for high‑temperature electrochemical systems requiring non‑volatile, thermally stable electrolytes
Extraction, Separation & Analytics
- Effective extractant for metal ions, organic molecules, and halogenated species in biphasic systems
- Supports liquid–liquid extraction, ion‑pairing separations, and hydrophobic partitioning studies
- Employed in analytical chemistry as an ion‑pairing agent, solvent, or electrolyte for electroanalytical methods
- Useful in chromatography method development where low‑volatility ionic media are required
Biomass Conversion & Green Chemistry
- Reaction medium for biomass dissolution, depolymerization, and catalytic upgrading
- Compatible with cellulose, lignin, and carbohydrate processing under mild or high‑temperature conditions
- Enables greener reaction pathways by replacing volatile organic solvents
Materials Science, Sensors & Soft‑Matter Interfaces
- Additive in ionogels, polymer electrolytes, and conductive soft materials
- Enhances sensitivity and stability in electrochemical sensors and flexible electronics
- Template or dispersant in nanoparticle synthesis, carbon materials, and hybrid composites
- Lubricant component for low‑friction, high‑load tribological systems
Please contact us if you want to learn more or need assistance with your order.