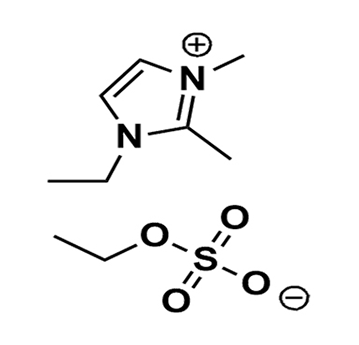
1-Ethyl-2,3-dimethylimidazolium ethyl sulfate >98%
Price range: $59.32 through $270.33
Product Code: IL-0207-HPCAS NO: 342573-75-5
- Chemical Formula: C9H18N2O4S
- Synonyms: EDiMIM EtSO4, Im112 EtSO4
- Hydrophilic
SUM Formula: C9H18N2O4S
Molecular Weight: 250.31
Purity: >98%
- SUM Formula: C9H18N2O4S
- Molecular Weight: 250.31
1-Ethyl-2,3-dimethylimidazolium ethyl sulfate, CAS: 342573-75-5
Key Applications:
Biomass & Biopolymer Processing
- Efficient dissolution and swelling of cellulose, hemicellulose, and lignocellulosic feedstocks for pretreatment and fractionation.
- Medium for carbohydrate derivatization, esterification, and selective depolymerization.
- Solvent for chitin/chitosan modification and homogeneous processing.
Acid‑Catalyzed Organic Transformations
- Reaction medium for Friedel–Crafts, esterification, acylation, and alkylation reactions where ethyl sulfate’s Brønsted acidity enhances catalytic turnover.
- Facilitates heterogeneous–homogeneous hybrid catalysis, improving mass transfer and selectivity.
- Useful in sulfonation and transesterification workflows requiring thermally stable, non‑volatile ionic media.
Electrochemical & Energy Applications
- High‑conductivity electrolyte component for aqueous and mixed‑solvent electrochemical systems.
- Additive in supercapacitor and pseudocapacitor formulations to improve ionic mobility and electrode wetting.
- Medium for electrodeposition of metals and metal oxides under mild conditions.
Surfactants, Formulations & Phase Behavior
- Hydrophilic IL for microemulsion formation, biphasic catalysis, and tunable solvent systems.
- Enhances solubility of polar organics and inorganic salts in green formulation chemistry.
- Compatible with enzyme‑assisted reactions, stabilizing certain biocatalysts in low‑water environments.
Thermal & Process Engineering
- Heat‑transfer and thermal‑storage medium in low‑temperature ranges due to high thermal stability and negligible vapor pressure.
- Solvent for extraction, ion‑exchange, and selective separation of polar compounds.
Please contact us if you want to learn more or need assistance with your order.