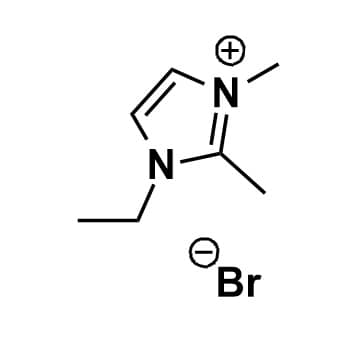
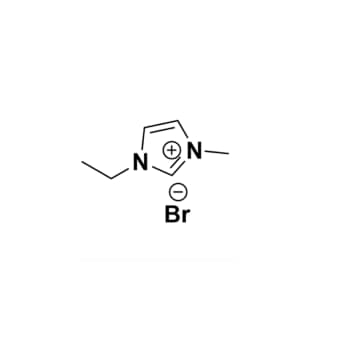
1-Ethyl-2,3-dimethylimidazolium bromide, >99%
Price range: $116.07 through $4,911.65
Product Code: IL-0135-HPCAS NO: 98892-76-3
- Chemical Fr.: C7H13BrN2
- Synonyms: EDiMIM Br, Im112 Br
- Aromatic
SUM Formula: C7H13BrN2
Molecular Weight: 205.10
Melting Point: 140 °C
Density: 1.468 g/cm³
Purity: >99%
- SUM Formula: C7H13BrN2
- Molecular Weight: 205.10
- Melting Point: 140 °C
- Density: 1.468 g/cm³
1-Ethyl-2,3-dimethylimidazolium bromide, CAS: 98892-76-3
Key Applications:
Catalysis & Organic Synthesis
- Functions as a polar, non‑volatile reaction medium for nucleophilic substitutions, oxidations, reductions, and C–C bond‑forming reactions.
- Bromide anion supports phase‑transfer catalysis, enhancing rates in biphasic organic–aqueous systems.
- Useful for transition‑metal‑catalyzed transformations, where steric hindrance around the imidazolium ring improves catalyst stability.
Electrochemical Systems
- Serves as a supporting electrolyte in electrochemical studies requiring high ionic conductivity and wide liquid‑phase stability.
- Employed in redox‑active systems, electrodeposition, and electroanalytical experiments where halide coordination is advantageous.
- The 2,3‑dimethyl substitution pattern reduces ring acidity, improving electrochemical stability windows relative to unsubstituted imidazoliums.
Biomolecular & Materials Solvation
- Effective solvent or co‑solvent for proteins, peptides, and nucleic acids, enabling controlled denaturation, refolding, or stabilization studies.
- Supports polymer dissolution and processing, particularly for charged or hydrogen‑bond‑rich polymers.
- Useful in nanomaterials synthesis, including templated growth of metal nanoparticles and ionic‑liquid‑mediated self‑assembly.
Thermal & Process Engineering
- High thermal stability and negligible vapor pressure make it suitable for high‑temperature reaction media and heat‑transfer environments.
- Acts as a green solvent alternative in processes seeking to reduce VOC emissions.
Surfactant, Micelle, and Soft‑Matter Systems
- Bromide counterion enables formation of ionic‑liquid‑based micelles, microemulsions, and structured fluids.
- Useful in colloid stabilization, nanoparticle dispersion, and interfacial engineering.
Precursor for Ionic‑Liquid Derivatives
- Serves as a synthetic intermediate for preparing hydrophobic or task‑specific ionic liquids via anion exchange (e.g., TFSI⁻, PF₆⁻, FSI⁻).
- Valuable for tuning viscosity, polarity, and hydrophobicity in custom IL formulations.
Please contact us if you want to learn more or need assistance with your order.