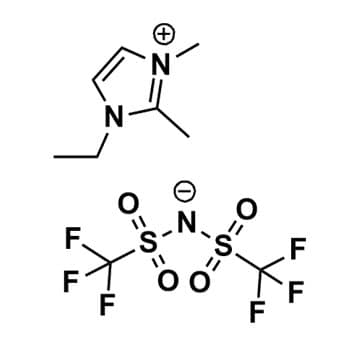
1-Ethyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $164.15 through $9,033.43
Product Code: IL-0106-HPCAS NO: 174899-90-2
- Chemical Formula: C9H13F6N3O4S2
- Synonyms: EDiMIM BTA, EDiMIM NTf2, EDiMIM TFSI, EDiMIM BTI, Im112 BTA
- Weakly-coordinating anion
- Aromatic
Conductivity: 3.18 mS/cm (20 °C)
SUM Formula: C9H13F6N3O4S2
Molecular Weight: 405.34
Melting Point: 25 °C
Density: 1.49 g/cm³ (25 °C)
ECW: 4.4 V
HMIS Key: NA
Purity: >99%
Viscosity: 68.5 cP (25 °C)
- SPECIFIC GRAVITY: NA
- SUM Formula: C9H13F6N3O4S2
- Molecular Weight: 405.34
- Melting Point: 25 °C
- Density: 1.49 g/cm³ (25 °C)
- ECW: 4.4 V
- HMIS KEY: NA
- TSCA: NA
- Viscosity: 68.5 cP (25 °C)
1-Ethyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 174899-90-2
Key Applications:
1. Electrochemical Systems
- Lithium‑ion and sodium‑ion electrolytes
- High oxidative stability supports high‑voltage cathodes.
- Hydrophobic TFSI⁻ anion improves SEI formation and reduces parasitic reactions.
- Supercapacitors and hybrid capacitors
- Low viscosity relative to bulkier imidazoliums improves ion mobility.
- Wide electrochemical window enables higher energy density.
- Electrodeposition media
- Stable under reductive conditions; supports smooth metal deposition (Al, Mg, rare‑earths).
2. Synthetic Chemistry & Catalysis
- Non‑nucleophilic reaction medium
- Excellent for strong electrophiles, Lewis acids, and moisture‑sensitive transformations.
- Organometallic catalysis
- Supports homogeneous catalysts requiring inert, non‑coordinating environments.
- High‑temperature synthesis
- Thermal stability of the TFSI⁻ anion allows reactions above 200 °C without decomposition.
3. Thermal & Process Engineering
- Heat‑transfer and thermal‑storage fluids
- High thermal stability and low vapor pressure make it suitable for closed‑loop systems.
- Solvent for high‑temperature separations
- Useful in extractive distillation and purification of aromatics or specialty organics.
4. Materials Science & Surface Engineering
- Polymer electrolytes and ion gels
- Enhances ionic conductivity while maintaining mechanical stability.
- Surface modification & lubrication
- Hydrophobic, low‑volatility IL for boundary lubrication in metal–metal contacts.
- Nanomaterial dispersion
- Stabilizes conductive nanomaterials (CNTs, graphene) due to π‑cation interactions.
5. Gas Capture & Separation
- SO₂, HF, and acidic gas absorption
- TFSI⁻ anion provides high chemical resistance; cation structure tunes solubility.
- Fluorinated gas separations
- Strong affinity for fluorinated species due to the perfluorinated anion.
6. Safety‑Critical & Harsh‑Environment Applications
- Non‑flammable electrolyte or solvent
- Suitable for aerospace, defense, and high‑temperature industrial systems.
- Radiation‑resistant media
- Imidazolium–TFSI systems show resilience under ionizing radiation, useful in nuclear‑adjacent environments.
Contact us if you want to learn more or need assistance with your order.