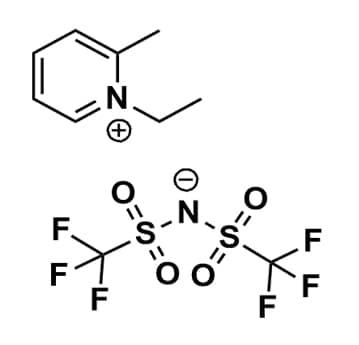
1-Ethyl-2-methylpyridinium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $202.34 through $11,340.61
Product Code: IL-0247-HPCAS NO: 712354-99-9
- Chemical Formula: C10H12F6N2O4S2
- Synonyms: 1-Ethyl-2-picolinium bis(trifluoromethylsulfonyl)imide, N-Ethyl-2-methylpyridinium bis(trifluoromethylsulfonyl)imide, Et2Pic BTA, Et-2-Pic BTA, Et2Pic NTf2, Et2Pic TFSI, Et2Pic BTI
- Weakly coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 3.71 mS/cm (30 °C)
SUM Formula: C10H12F6N2O4S2
Molecular Weight: 402.33
Melting Point: <RT
Density: 1.51 g/cm³ (27 °C)
ECW: 4.4 V
Purity: >99%
Viscosity: 74.8 cP (25 °C)
- SUM Formula: C10H12F6N2O4S2
- Molecular Weight: 402.33
- Melting Point:
- Density: 1.51 g/cm³ (27 °C)
- ECW: 4.4 V
- Viscosity: 74.8 cP (25 °C)
1-Ethyl-2-methylpyridinium bis(trifluoromethylsulfonyl)imide, CAS: 712354-99-9
Key Applications:
Electrochemical Systems
- High‑stability electrolyte component for lithium, sodium, and multivalent ion batteries where low viscosity and high ionic conductivity are required.
- Additive for high‑voltage electrolyte formulations, improving oxidative stability and suppressing parasitic reactions at electrode interfaces.
- Electrolyte medium for supercapacitors, particularly in devices requiring wide electrochemical windows and low volatility.
- Solvent or co‑solvent in electrodeposition baths, supporting uniform metal deposition and improved current efficiency.
Thermal & Chemical Stability Applications
- Heat‑transfer and thermal‑management fluids in systems requiring non‑flammability and high thermal stability.
- Reaction medium for high‑temperature or strongly electrophilic transformations, leveraging the TFSI anion’s exceptional chemical inertness.
- Solvent for catalytic processes, especially those involving transition‑metal complexes that benefit from weakly coordinating anions.
Separation Science & Extraction
- Phase‑transfer medium for metal ion extraction, including rare‑earth and precious‑metal separations.
- Selective solvent for hydrophobic analytes in liquid–liquid extraction workflows.
- Stationary‑phase modifier in chromatography, enhancing retention and selectivity for nonpolar or aromatic species.
Materials Science & Polymer Processing
- Plasticizer or processing aid for high‑performance polymers, improving flexibility and ionic mobility.
- Doping medium for conductive polymers such as PEDOT derivatives, enhancing conductivity and mechanical stability.
- Template or structuring solvent in the synthesis of porous materials, ionogels, and hybrid organic–inorganic matrices.
Surface & Interface Engineering
- Surface‑modification medium for tuning wettability, charge density, or interfacial adhesion on metals, oxides, and carbon materials.
- Electrochemical cleaning or passivation baths, benefiting from the ionic liquid’s low volatility and broad electrochemical window.
Analytical & Spectroscopic Uses
- Matrix or solvent for spectroscopic studies (NMR, IR, Raman) where low vapor pressure and high thermal stability are advantageous.
- Medium for studying ion–molecule interactions, proton transfer, and solvation dynamics due to its asymmetric pyridinium cation.
Please contact us if you want to learn more or need assistance with your order.