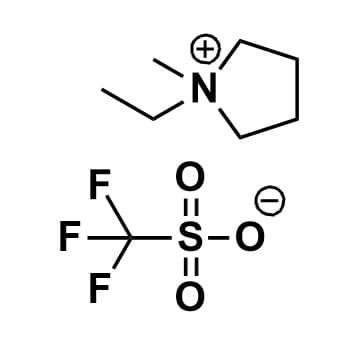
1-Ethyl-1-methylpyrrolidinium triflate, >99%
Price range: $314.22 through $3,466.39
Product Code: IL-0168-HPCAS NO: 8893443-18-0
- Chemical Formula: C8H16F3NO3S
- Synonyms: 1-Ethyl-1-methylpyrrolidinium trifluoromethanesulfonate, N-Ethyl-N-methylpyrrolidinium trifluoromethanesulfonate, EMPyrr OTf, PYR12 OTf, PY12 OTf
SUM Formula: C8H16F3NO3S
Molecular Weight: 263.28
Melting Point: 107 °C
Density: 1.337 g/cm³
Purity: >98%
- SUM Formula: C8H16F3NO3S
- Molecular Weight: 263.28
- Melting Point: 107 °C
- Density: 1.337 g/cm³
1-Ethyl-1-methylpyrrolidinium triflate, CAS: 8893443-18-0
Key Applications
- Electrolytes for Energy Storage Systems
Used as a high‑conductivity ionic component in lithium‑ion, sodium‑ion, and emerging multivalent battery systems. The combination of a stable pyrrolidinium cation and a weakly coordinating triflate anion supports wide electrochemical windows, reduced flammability, and improved thermal stability in both liquid and gel polymer electrolytes.
- Supercapacitors and Hybrid Capacitors
Incorporated into ionic liquid–based electrolytes to enhance double‑layer capacitance, extend operational voltage, and improve cycling durability. Its low viscosity relative to bulkier ionic liquids enables faster ion transport in porous carbon electrodes.
- Electrochemical Devices and Sensors
Serves as a supporting electrolyte in voltammetric and amperometric systems where high ionic strength, low volatility, and chemical inertness are required. Suitable for microfabricated sensors, high‑temperature electrochemical cells, and ionic‑liquid‑modified electrodes.
- Catalysis and Organic Synthesis
Functions as a non‑nucleophilic ionic medium for acid‑catalyzed, transition‑metal‑catalyzed, and electrophilic transformations. The triflate anion provides enhanced solubility for organometallic complexes and can stabilize reactive intermediates in selective transformations.
- Separation Science and Chromatography
Applied as an ionic liquid additive in HPLC and CE to tune selectivity, suppress analyte adsorption, and improve peak shape. The pyrrolidinium cation offers strong elution‑modifying effects without introducing UV‑active impurities.
- Polymer Processing and Ionogel Formation
Used in the fabrication of ionogels, polymer electrolytes, and conductive elastomers. Its thermal stability and compatibility with acrylates, urethanes, and fluoropolymers support mechanically robust, ionically conductive materials.
- CO₂ Capture and Gas Solvation Studies
Employed in research on gas solubility, ionic‑liquid‑based absorption media, and transport modeling. The triflate anion’s moderate coordinating strength enables predictable solvation behavior for CO₂ and other small gases.
- Electrodeposition and Surface Engineering
Acts as a stable ionic medium for metal deposition processes requiring low water content and high electrochemical stability. Suitable for controlled deposition of metals and alloys under anhydrous conditions.
Please contact us if you want to learn more or need assistance with your order.