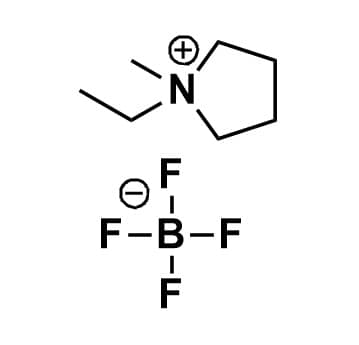
1-Ethyl-1-methylpyrrolidinium tetrafluoroborate, >99%
Price range: $445.86 through $2,483.93
Product Code: IL-0165-UPCAS NO: 117947-85-0
- Chemical Fr: C7H16BF4N
- Synonyms: EMPyrr BF4, PYR12 BF4, PY12 BF4, N-Ethyl-N-methylpyrrolidinium tetrafluoroborate
- Anion decomposes slowly in the presence of water
- Weakly coordinated anion
- Hydrophobic
For High-Purity of >98% refer to IL-0165-HP
SUM Formula: C7H16BF4N
Molecular Weight: 201.01
Melting Point: 292 °C
Purity: >99%
- SUM Formula: C7H16BF4N
- Molecular Weight: 201.01
- Melting Point: 292 °C
1-Ethyl-1-methylpyrrolidinium tetrafluoroborate, CAS: 117947-85-0
Key Applications:
Electrolytes and Energy Storage
- Serves as a stable ionic liquid electrolyte component for lithium‑ion, sodium‑ion, and emerging multivalent battery systems.
- Offers wide electrochemical windows and low volatility, supporting high‑temperature and high‑voltage cell designs.
- Used in supercapacitor electrolytes to enhance ionic conductivity and thermal robustness.
Electrodeposition and Surface Engineering
- Functions as an additive or primary medium for metal electrodeposition, particularly where smooth, uniform films are required.
- Supports controlled deposition of metals such as copper, nickel, and tin in non‑aqueous environments.
- Applied in surface finishing workflows that demand low water content and high chemical stability.
Catalysis and Organic Synthesis
- Acts as a non‑volatile, thermally stable solvent for transition‑metal‑catalyzed reactions.
- Enables selective transformations in nucleophilic substitutions, oxidations, and coupling reactions under anhydrous conditions.
- Useful in biphasic catalysis where ionic liquids facilitate catalyst recovery and reuse.
Separation Science and Extraction
- Employed in liquid–liquid extraction systems for selective separation of metal ions and organic species.
- Supports chromatographic applications requiring tunable polarity and low vapor pressure.
- Utilized in solvent‑free or reduced‑solvent purification workflows.
Materials Processing and Polymer Science
- Serves as a processing aid for polymer dissolution, dispersion, and ion transport modification.
- Integrated into polymer electrolytes and ion‑conducting films to improve mechanical flexibility and conductivity.
- Used in formulation of specialty coatings requiring electrochemical stability.
Electrochemical Devices and Sensors
- Provides a stable medium for redox‑active species in electrochemical sensors.
- Enhances signal stability in high‑temperature or moisture‑sensitive sensing environments.
- Supports development of ionic‑liquid‑based reference systems.
Please contact us if you want to learn more or need assistance with your order.