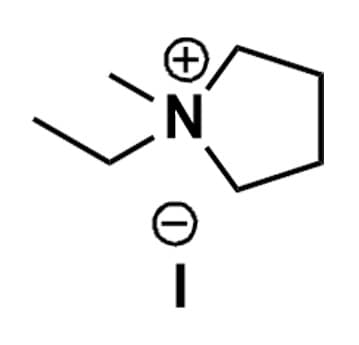
1-Ethyl-1-methylpyrrolidinium iodide, >98%
Price range: $236.34 through $2,600.13
Product Code: IL-0164-HPCAS NO: 4186-68-9
- Chemical Formula: C7H16IN
- Synonyms: EMPyrr I, PYR12 I, PY12 I, N-Ethyl-N-methylpyrrolidinium iodide
- Light sensitive
SUM Formula: C7H16IN
Molecular Weight: 241.11
Melting Point: >300 °C
Density: 1.470 g/cm³ (25 °C)
Purity: >98%
- SUM Formula: C7H16IN
- Molecular Weight: 241.11
- Melting Point: >300 °C
- Density: 1.470 g/cm³ (25 °C)
1-Ethyl-1-methylpyrrolidinium iodide, CAS: 4186-68-9
Key Applications:
Electrochemical Systems
- Serves as a quaternary ammonium iodide salt for use in electrolyte formulation, particularly where iodide anions support redox mediation or halide exchange.
- Utilized in reference electrolyte studies to evaluate cation–anion interactions, ionic mobility, and conductivity trends within pyrrolidinium-based salts.
- Supports electrode interface investigations, including adsorption behavior and halide-driven surface modification.
Materials Science and Ion Transport Studies
- Employed as a model ionic species in transport, viscosity, and ion-pairing research, enabling comparison across pyrrolidinium iodide derivatives.
- Useful in solid–liquid interface studies, including polymer–salt compatibility, ion dissociation behavior, and dielectric response mapping.
- Incorporated into polymer electrolyte matrices to probe mechanical and conductive properties influenced by bulky organic cations.
Catalysis and Synthesis
- Functions as a phase-transfer catalyst or halide source in organic transformations requiring iodide-mediated nucleophilicity.
- Supports halide exchange reactions, enabling controlled introduction of iodide into organometallic or coordination complexes.
- Applied in ionic-liquid-assisted synthesis, where the cation structure provides tunable solvation and microenvironment effects.
Photochemical and Redox Applications
- Utilized in iodide/triiodide redox couple studies, including dye-sensitized solar cell research and photoredox benchmarking.
- Suitable for charge-transfer investigations, where iodide’s redox activity and the pyrrolidinium cation’s stability enable reproducible measurements.
Analytical and Spectroscopic Uses
- Serves as a reference iodide salt for calibration, ion-pairing studies, and comparative NMR, FTIR, or Raman analyses.
- Supports solvation and ion-association research, particularly in mixed-solvent systems where cation structure influences iodide behavior.
General Research Utility
- Provides a stable, non-volatile iodide source for halide chemistry, ionic liquid precursor studies, and structure–property mapping across quaternary ammonium salts.
- Suitable for method development in electrochemical, spectroscopic, and materials characterization workflows.
Please contact us if you want to learn more or if you need assistance with your order.