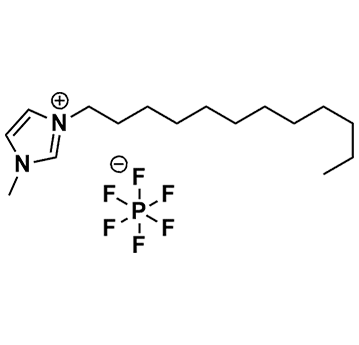
1-Dodecyl-3-methylimidazolium hexafluorophosphate, >98%
Price range: $192.46 through $10,722.07
Product Code: IL-0095-HPCAS NO: 219947-93-0
- Chemical Formula: C16H31F6N2P
- Synonyms: DodecMIM PF 6, C 1 C 12 Im PF 6, Im 1.12 PF 6
- Anion decomposes slowly in the presence of water.
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
SUM Formula: C16H31F6N2P
Molecular Weight: 396.96
Melting Point: 60°C
Density: 0.971 g/cm³
Purity: >98%
- SUM Formula: C16H31F6N2P
- Molecular Weight: 396.96
- Melting Point: 60°C
- Density: 0.971 g/cm³
1-Dodecyl-3-methylimidazolium hexafluorophosphate, CAS: 219947-93-0
Key Applications:
Solvent and Co‑solvent in Organic Synthesis
- Supports reactions requiring low‑volatility, thermally stable media.
- Useful in nucleophilic substitutions, phase‑transfer‑assisted transformations, and moisture‑sensitive processes.
- Enhances solubility of long‑chain organics and hydrophobic intermediates.
Electrochemical and Ionic Conductivity Applications
- Serves as a hydrophobic ionic liquid electrolyte with good electrochemical stability.
- Applied in electrodeposition studies, redox‑active systems, and ion‑transport investigations.
- Suitable for evaluating interfacial behavior at liquid–liquid and liquid–solid boundaries.
Surfactant, Self‑Assembly, and Soft‑Matter Systems
- Long C12 alkyl chain promotes micelle formation, vesicle assembly, and ordered nanostructures.
- Used in templating studies for mesoporous materials and controlled‑morphology nanomaterials.
- Supports investigations into amphiphilic ionic liquid behavior and critical micelle concentration (CMC) tuning.
Separation Science and Extraction
- Effective as an extractant or phase modifier in biphasic ionic liquid systems.
- Applied in metal ion extraction, hydrophobic analyte partitioning, and solvent‑free purification strategies.
- Enhances selectivity in liquid–liquid extraction due to its hydrophobic PF6– anion.
Materials Science and Thin‑Film Fabrication
- Used in polymer–ionic liquid composites to improve flexibility, conductivity, and thermal stability.
- Supports fabrication of ion‑conductive membranes and coatings.
- Useful in modifying surface charge, wettability, and interfacial adhesion.
Analytical Chemistry and Spectroscopic Studies
- Provides a stable, low‑volatility medium for NMR, UV‑Vis, and fluorescence studies.
- Enables investigation of solvation dynamics, ion pairing, and microenvironment polarity.
- Suitable for calibrating hydrophobic ionic liquid behavior in method development.
Catalysis and Reaction Engineering
- Functions as a non‑volatile reaction medium for acid‑catalyzed and metal‑catalyzed transformations.
- Can enhance catalyst dispersion and turnover in biphasic catalytic systems.
- Supports recyclable catalyst platforms due to its low vapor pressure and phase‑separation behavior.
Please contact us if you want to learn more or need assistance with your order.