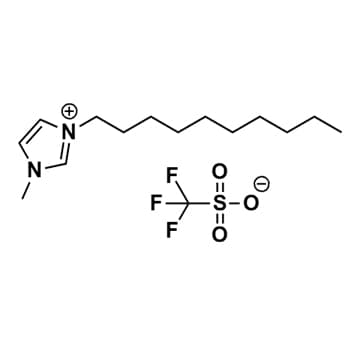
1-Decyl-3-methylimidazolium triflate, >99%
Price range: $267.49 through $3,838.71
Product Code: IL-0068-HPCAS NO: 412009-62-2
- Chemical Formula: C15H27F3N2O3S
- Synonyms: DecMIM OTf, C1C10Im OTf, Im1,10 OTf, 1-Decyl-3-methylimidazolium trifluoromethylsulfonate
- Hydrophobic
- Aromatic
Conductivity: 0.41 mS/cm (24 °C)
SUM Formula: C15H27F3N2O3S
Molecular Weight: 372.45
Melting Point: 27 °C
Density: 1.15 g/cm³ (26 °C)
Purity: >99%
Viscosity: 298 cP (25 °C)
- SUM Formula: C15H27F3N2O3S
- Molecular Weight: 372.45
- Melting Point: 27 °C
- Density: 1.15 g/cm³ (26 °C)
- Viscosity: 298 cP (25 °C)
1-Decyl-3-methylimidazolium triflate, CAS: 412009-62-2
Key Applications:
Solvent and Medium for Organic Synthesis
- Supports reactions requiring low‑volatility, thermally stable media.
- Useful in alkylation, nucleophilic substitution, and acid‑catalyzed transformations where the triflate anion provides enhanced ionic conductivity and moderate coordinating ability.
- Enables biphasic or phase‑transfer‑like systems due to its amphiphilic C10 chain.
Electrochemical and Ionic Conductivity Applications
- Functions as a high‑conductivity electrolyte component in electrochemical cells, especially where hydrophobicity and wide electrochemical windows are desired.
- Suitable for electrodeposition studies and ionic‑liquid‑based capacitors where long‑chain imidazolium cations improve interfacial structuring.
Materials Processing and Polymer Science
- Acts as a plasticizing or ion‑conducting additive in polymer matrices, including PVDF, PMMA, and ionomer systems.
- Enhances flexibility, ionic mobility, and thermal stability in polymer electrolytes and membranes.
- Useful in templating or directing nanostructure formation in block copolymers due to its amphiphilic character.
Surfactant‑Like and Self‑Assembly Behavior
- The decyl chain imparts strong amphiphilicity, enabling micelle‑like aggregation in certain solvents.
- Supports formation of ordered soft materials, thin films, and structured liquids.
- Useful in dispersing nanoparticles or stabilizing colloidal systems where ionic surface interactions are required.
Catalysis and Phase Behavior Engineering
- Serves as a tunable medium for acid‑catalyzed and metal‑catalyzed reactions, leveraging the weakly coordinating triflate anion.
- Facilitates catalyst recycling and separation due to its low volatility and immiscibility with many organic solvents.
- Enables temperature‑dependent phase behavior useful for reaction control and product isolation.
Extraction, Separation, and Partitioning
- Effective in liquid–liquid extraction systems where hydrophobic ionic liquids enhance selectivity for aromatic, sulfur‑containing, or metal‑ion species.
- The long alkyl chain improves partitioning behavior and reduces miscibility with aqueous phases.
Nanomaterials and Surface Modification
- Useful as a stabilizing medium for metal nanoparticles, carbon nanomaterials, and metal oxides.
- Enhances dispersion quality and surface charge control in hydrophobic or mixed‑polarity environments.
Please contact us if you want to learn more or need assistance with your order.