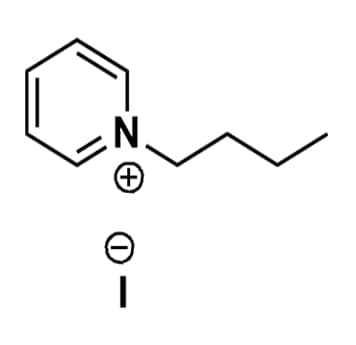
1-Butylpyridinium iodide, >98%
Price range: $260.45 through $2,053.78
Product Code: IL-0176-HPCAS NO: 874-81-7
- Chemical Formula: C9H14IN
- Synonyms: BuPy I, N-Butylpyridinium iodide
- Light sensitive
- Aromatic
SUM Formula: C9H14IN
Molecular Weight: 263.12
Melting Point: 74 °C
Density: 1.506 g/cm³ (25 °C)
Purity: >98%
- SUM Formula: C9H14IN
- Molecular Weight: 263.12
- Melting Point: 74 °C
- Density: 1.506 g/cm³ (25 °C)
1-Butylpyridinium iodide, CAS: 874-81-7
Key Applications:
Electrochemistry and Charge‑Transfer Systems
- Functions as a supporting electrolyte in non‑aqueous electrochemical studies where a bulky, stable pyridinium cation is preferred.
- Useful in redox‑active systems requiring high ionic strength and low volatility.
- Supports ion‑transport investigations in mixed organic solvents and ionic‑liquid precursor matrices.
Precursor for Ionic Liquid and Salt Design
- Serves as a quaternary ammonium building block for synthesizing task‑specific ionic liquids via anion exchange.
- Enables formulation of halide‑rich ionic environments for catalysis, separations, and materials processing.
Phase‑Transfer and Halide‑Mediated Catalysis
- Acts as a phase‑transfer catalyst in organic transformations involving nucleophilic substitution, alkylation, and halide‑driven mechanisms.
- Provides controlled iodide delivery in reactions where iodide acts as a soft nucleophile or catalytic promoter.
Solvent Extraction and Ion‑Pairing Studies
- Supports extraction systems where iodide‑based ion pairing enhances selectivity for soft metal ions (e.g., Ag⁺, Pd²⁺).
- Useful in studying ion‑association equilibria in low‑polarity media.
Materials Science and Thin‑Film Processing
- Employed as an additive or templating agent in the preparation of hybrid organic–inorganic materials.
- Supports halide‑rich environments for perovskite precursor studies, crystallization behavior, and film morphology control.
Spectroscopy and Analytical Chemistry
- Provides a well‑defined pyridinium reference ion for NMR, FTIR, and conductivity studies.
- Useful in probing ion–solvent interactions and halide coordination behavior.
General Research Utility
- Suitable for halide‑exchange experiments, nucleophilicity comparisons, and benchmarking studies across pyridinium salt families.
- Offers a stable, high‑purity (>98%) iodide source for method development in synthetic and physical chemistry.
Please contact us if you want to learn more or need assistance with your order.