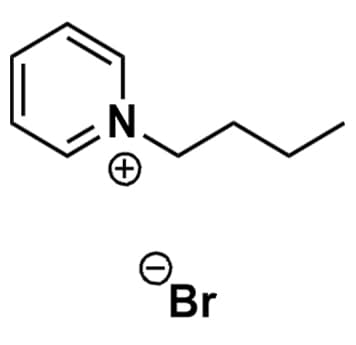
1-Butylpyridinium bromide, >99%
Price range: $54.18 through $5,058.87
Product Code: IL-0086-HPCAS NO: 874-80-6
- Chemical Formula: C9H14BrN
- Synonyms: N-Butylpyridinium bromide, BuPy Br
- Aromatic
SUM Formula: C9H14BrN
Molecular Weight: 216.12
Melting Point: 98 °C
Purity: >99%
- SUM Formula: C9H14BrN
- Molecular Weight: 216.12
- Melting Point: 98 °C
1-Butylpyridinium bromide, CAS: 874-80-6
Key Applications:
Electrochemistry & Charge‑Transfer Studies
- Serves as a supporting electrolyte in voltammetry, impedance spectroscopy, and redox‑active material testing.
- Enhances ionic conductivity in non‑aqueous systems and stabilizes electrode–electrolyte interfaces.
- Useful in evaluating ion‑pairing effects and transport behavior in model electrolyte systems.
Phase‑Transfer & Organic Synthesis
- Functions as a phase‑transfer catalyst for nucleophilic substitution, alkylation, and heterocycle formation.
- Facilitates reactions in biphasic systems where enhanced solubilization of ionic intermediates is required.
- Supports mechanistic studies on pyridinium‑based catalytic environments.
Solvent & Extraction Systems
- Acts as a tunable ionic medium for liquid–liquid extraction of metal ions, dyes, and organic contaminants.
- Useful in designing task‑specific ionic liquid systems for selective separations.
- Supports studies on solvation dynamics, polarity tuning, and ion‑exchange behavior.
Materials Science & Polymer Research
- Employed as a structure‑directing additive in polymer electrolytes, ion‑conducting films, and hybrid composites.
- Enhances mechanical and thermal properties in polymer matrices through ionic cross‑linking interactions.
- Used in templating and morphology control for nanostructured materials.
Surfactant & Self‑Assembly Studies
- Supports research on micelle formation, critical aggregation behavior, and amphiphilic ionic systems.
- Useful in probing interfacial phenomena and adsorption at solid–liquid boundaries.
- Enables formulation of ionic surfactant systems for controlled dispersion and stabilization.
Analytical & Spectroscopic Applications
- Provides a well‑defined ionic environment for NMR, UV–Vis, and IR studies of ion pairing and solvation.
- Used as a calibration or reference salt in studies of ionic strength and conductivity.
Please contact us if you want to learn more or need assistance with your order.