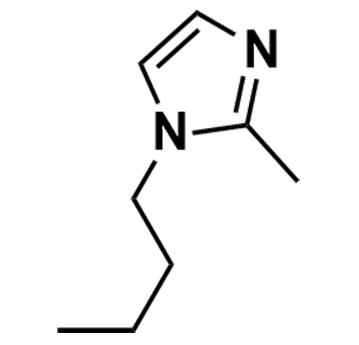
1-Butylimidazole >98%
Price range: $202.48 through $2,203.91
Product Code: KI-0004-HPCAS NO: 4316-42-1
- Chemical Formula:C7H12N2
- Synonyms: BuIM
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C7H12N2
Molecular Weight: 124.18
Purity: >98%
- SPECIFIC GRAVITY: NA
- SUM Formula: C7H12N2
- Molecular Weight: 124.18
- TSCA: NA
1-Butylimidazole, CAS: 4316-42-1
Key Applications:
Ionic Liquids and Functional Salts
- Precursor for synthesizing 1‑butyl‑substituted imidazolium ionic liquids with halide, BF₄⁻, PF₆⁻, NTf₂⁻, acetate, and other anions.
- Supports development of task‑specific ionic liquids for electrochemistry, separations, solvent engineering, and electrolyte formulation.
Catalysis and Coordination Chemistry
- Nitrogen‑donor ligand for preparing metal complexes used in homogeneous catalysis.
- Applied in catalytic systems for alkylation, esterification, polymer modification, and fine‑chemical synthesis.
Organic Synthesis Intermediate
- Nucleophilic heterocycle for quaternization, N‑alkylation, and heterocycle functionalization.
- Intermediate for surfactants, specialty intermediates, and heterocyclic materials.
Electrolyte and Energy‑Materials Development
- Starting material for ionic‑liquid electrolytes in batteries, supercapacitors, and electrochemical devices.
- Enables tuning of viscosity, conductivity, and thermal stability through controlled cation design.
Corrosion Inhibitor Research
- Imidazole derivatives, including butyl‑substituted analogs, are evaluated as corrosion‑inhibiting agents for steel and copper systems.
- Supports formulation studies in coatings, lubricants, and industrial water treatment.
Polymer and Materials Modification
- Introduces imidazole functionality into polymer backbones or side chains.
- Used in ion‑conductive polymers, crosslinkable materials, and advanced coatings.
Pharmaceutical Research
- Heterocycle precursor for synthesizing imidazolium‑based intermediates with antimicrobial, antifungal, and antiparasitic relevance.
- Building block for ionic‑liquid‑based drug delivery systems designed to modulate solubility and permeability.
- Synthetic auxiliary in N‑alkylation and quaternization reactions for generating bioactive imidazolium scaffolds.
- Ligand component in catalytic transformations used in API synthesis.
Agrochemical Development
- Intermediate for quaternary imidazolium salts evaluated as plant‑protection agents, growth regulators, and antimicrobial additives.
- Precursor for ionic‑liquid‑based extraction systems used in isolating natural products, pesticide metabolites, and soil‑bound residues.
- Functional heterocycle incorporated into candidate herbicidal and fungicidal molecules.
- Catalyst/solvent component in green‑chemistry synthesis routes for agrochemical actives and formulation additives.
Please contact us if you want to learn more or need assistance with your order.