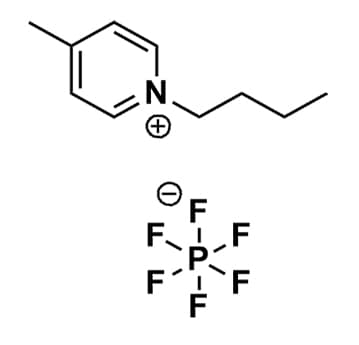
1-Butyl-4-methylpyridinium hexafluorophosphate, >99%
Price range: $199.50 through $8,639.98
Product Code: IL-0084-HPCAS NO: 401788-99-6
- Chemical Formula: C10H16F6NP
- Synonyms: Bu-4-Pic PF6, Bu4Pic PF6, 1-Butyl-4-picolinium hexafluorophosphate, N-Butyl-4-methylpyridinium hexafluorophosphate, N-Butyl-4-picolinium hexafluorophosphate
- Anion decomposes slowly in the presence of water.
- Weakly-Coordination Anion
- Hydrophobic
- Aromatic
SUM Formula: C10H16F6NP
Molecular Weight: 295.21
Melting Point: 42–43 °C
Density: 1.327 g/cm³ (25 °C)
Purity: >99%
- SUM Formula: C10H16F6NP
- Molecular Weight: 295.21
- Melting Point: 42–43 °C
- Density: 1.327 g/cm³ (25 °C)
1-Butyl-4-methylpyridinium hexafluorophosphate, CAS: 401788-99-6
Key Applications:
Electrochemical Systems
- Serves as a high‑stability ionic liquid electrolyte for supercapacitors, redox‑flow systems, and advanced battery research.
- Offers wide electrochemical windows and low volatility, supporting high‑voltage device development.
- Useful as a supporting electrolyte in cyclic voltammetry and electrodeposition studies where PF₆⁻ stability is advantageous.
Catalysis and Synthesis
- Functions as a non‑volatile reaction medium for transition‑metal‑catalyzed transformations, including cross‑coupling and selective alkylation.
- Enhances reaction rates and selectivity in biphasic and solvent‑free systems due to its polarity and thermal stability.
- Supports ionic‑liquid‑assisted synthesis of nanomaterials, enabling controlled nucleation and particle morphology.
Separation Science
- Applied in liquid–liquid extraction of metal ions, particularly for hydrophobic or soft‑acid species.
- Useful in chromatographic method development as an ionic liquid additive to tune retention, peak shape, and selectivity.
- Supports membrane‑based separations where ionic liquid incorporation improves ion transport and chemical resistance.
Materials and Surface Engineering
- Employed as a templating or structuring agent in the fabrication of porous materials, conductive films, and polymer composites.
- Enhances dispersion and interfacial compatibility in polymer matrices due to its amphiphilic cation and hydrophobic anion.
- Utilized in electrodeposition baths to improve coating uniformity and suppress dendritic growth.
Thermal and Chemical Stability Applications
- Suitable for high‑temperature reaction environments due to its low vapor pressure and robust PF₆⁻ anion.
- Acts as a heat‑transfer or thermal‑storage medium in specialized research settings requiring non‑flammable ionic liquids.
Solvation and Spectroscopy
- Provides a tunable, non‑coordinating solvent environment for spectroscopic studies of organometallic complexes and reactive intermediates.
- Supports solvatochromic and ion‑pairing investigations where controlled polarity and ionic strength are required.
Please contact us if you want to learn more or need assistance with your order.