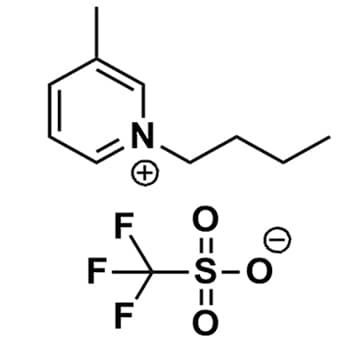
1-Butyl-3-methylpyridinium triflate, >98%
Price range: $591.59 through $4,757.39
Product Code: IL-0179-HPCAS NO: 857841-32-8
- Chemical Formula: C11H16F3NO3S
- Synonyms: Bu-3-Pic OTf, Bu3Pic OTf, 1-Butyl-3-methylpyridinium trifluoromethanesulfonate, 1-Butyl-3-picolinium triflate, N-Butyl-3-methylpyridinium triflate, N-Butyl-3-picolinium triflate
- Hydrophoic
- Aromatic
Conductivity: 2.35 mS/cm (30 °C)
SUM Formula: C11H16F3NO3S
Molecular Weight: 237.05
Melting Point: <RT
Density: 1.28 g/cm³ (25 °C)
Purity: >98%
Viscosity: 117 cP (25 °C)
- SUM Formula: C11H16F3NO3S
- Molecular Weight: 237.05
- Melting Point:
- Density: 1.28 g/cm³ (25 °C)
- Viscosity: 117 cP (25 °C)
1-Butyl-3-methylpyridinium triflate, CAS: 857841-32-8
Key Applications:
Electrochemical Systems
- Serves as a stable ionic medium for electrochemical studies requiring wide electrochemical windows and low volatility.
- Supports ion transport in prototype electrolytes for capacitors, redox systems, and electrodeposition research.
- Useful in evaluating ionic‑liquid–based interfaces due to its thermal stability and predictable conductivity profile.
Catalysis and Organic Synthesis
- Functions as a non‑volatile reaction medium for acid‑catalyzed and transition‑metal‑catalyzed transformations.
- Provides enhanced solvation for polar intermediates, enabling rate improvements in selected coupling, alkylation, and rearrangement reactions.
- Suitable for biphasic catalysis where phase separation and product recovery are important.
Materials Science and Polymer Processing
- Acts as a processing aid or plasticizing ionic component in polymer blends, ionogels, and hybrid materials.
- Supports the formation of conductive films and membranes where ionic mobility and thermal robustness are required.
- Enables dispersion and stabilization of nanoparticles in non‑aqueous matrices.
Separation Science
- Employed as a tunable solvent or co‑solvent in liquid–liquid extraction systems targeting metal ions, organics, and specialty analytes.
- Useful in chromatographic method development where ionic‑liquid additives improve peak shape, selectivity, or retention control.
Thermal and Transport Studies
- Provides a model system for studying ion–ion and ion–solvent interactions due to its well‑defined cation/anion pairing.
- Used in research on viscosity, conductivity, and diffusion behavior of pyridinium‑based ionic liquids.
Specialty Applications
- Supports development of ionic‑liquid–based lubricants and anti‑wear additives for high‑temperature environments.
- Investigated in CO₂ capture and gas‑solvation studies where triflate‑based ionic liquids offer predictable physicochemical behavior.
Please contact us if you want to learn more or need assistance with your order.