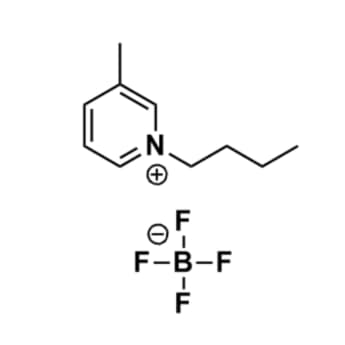
1-Butyl-3-methylpyridinium tetrafluoroborate, >99%
Price range: $190.97 through $8,305.86
Product Code: IL-0081-HPCAS NO: 597581-48-1
- Chemical Formula: C10H16BF4N
- Synonyms: Bu-3-Pic BF4, Bu3Pic BF4, 1-Butyl-3-picolinium tetrafluoroborate, N-Butyl-3-methylpyridinium tetrafluoroborate, N-Butyl-3-picolinium tetrafluoroborate
- Anion decomposes slowly in the presence of water
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 1.41 mS/cm (23 °C)
SUM Formula: C10H16BF4N
Molecular Weight: 237.05
Melting Point: <RT
Density: 1.18 g/cm³ (25 °C)
ECW: 3.7 V
Purity: >99%
Viscosity: 176 cP (25 °C)
- SUM Formula: C10H16BF4N
- Molecular Weight: 237.05
- Melting Point:
- Density: 1.18 g/cm³ (25 °C)
- ECW: 3.7 V
- Viscosity: 176 cP (25 °C)
1-Butyl-3-methylpyridinium tetrafluoroborate, CAS: 597581-48-1
Key Applications:
Electrochemical Systems
- High‑stability ionic liquid electrolyte with a wide liquid‑phase window and low volatility.
- Suitable for capacitors, dye‑sensitized solar cells, redox systems, and non‑aqueous electrochemical studies.
- Enables controlled electrodeposition processes where moisture‑free, low‑vapor‑pressure media are required.
Solvent and Co‑Solvent in Organic Synthesis
- Functions as a non‑volatile, tunable solvent for catalytic transformations and separation processes.
- Exhibits well‑characterized behavior in binary mixtures (e.g., with ethanol), supporting process modeling and thermodynamic design.
- Provides selective dissolution of polar and moderately non‑polar substrates.
Materials Science and Thin‑Film Processing
- Applied in ionic‑liquid templating, polymer doping, and nanomaterial dispersion.
- Supports thin‑film fabrication for optoelectronic materials due to controlled viscosity and thermal stability.
Thermophysical Property Benchmarking
- Extensively characterized in NIST Web Thermo Tables, making it a reference ionic liquid for density, viscosity, heat‑capacity, and enthalpy studies.
- Used in computational chemistry validation, including COSMO‑RS, molecular dynamics, and equation‑of‑state development.
Extraction and Separation Processes
- Serves as a task‑specific extraction medium for metal ions, aromatic compounds, and biomass‑derived molecules.
- BF₄⁻‑based ionic liquids demonstrate favorable partitioning behavior for selective separations.
Antistatic and Charge‑Control Additives
- Incorporated into coatings, lubricants, and polymer matrices to enhance ionic conductivity and reduce static buildup.
- Suitable for precision manufacturing environments requiring thermally stable, low‑volatility additives.
Please contact us if you want to learn more or need assistance with your order.