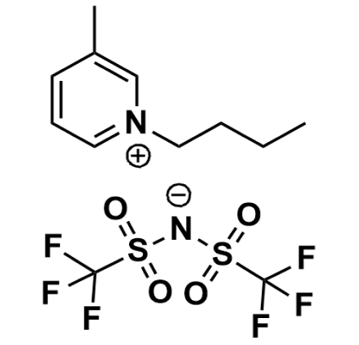
1-Butyl-3-methylpyridinium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $164.15 through $9,033.43
Product Code: IL-0216-HPCAS NO: 344790-86-9
- Chemical Formula: C12H16F6N2O4S2
- Synonyms: 1-Butyl-3-picolinium bis(trifluoromethylsulfonyl)imide, N-Butyl-3-methylpyridinium bis(trifluoromethylsulfonyl)imide, N-Butyl-3-picolinium bis(trifluoromethylsulfonyl)imide, Bu3Pic BTA, Bu-3-Pic BTA, Bu3Pic NTf2, Bu3Pic TFSI, Bu3Pic BTI
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 2.51 mS/cm (25 °C)
SUM Formula: C12H16F6N2O4S2
Molecular Weight: 430.39
Melting Point: <RT
Density: 1.41 g/cm³ (22 °C)
Purity: >99%
Viscosity: 51.8 cP (25 °C)
- SUM Formula: C12H16F6N2O4S2
- Molecular Weight: 430.39
- Melting Point:
- Density: 1.41 g/cm³ (22 °C)
- Viscosity: 51.8 cP (25 °C)
1-Butyl-3-methylpyridinium bis(trifluoromethylsulfonyl)imide, CAS: 344790-86-9
Key Applications
Electrochemical Systems
- Serves as a high‑stability electrolyte or electrolyte additive in lithium, sodium, and multivalent battery research, particularly where low volatility and high oxidative stability are required.
- Enables stable electrode–electrolyte interfaces in supercapacitors and hybrid capacitors due to its broad electrochemical window.
- Functions as a solvent medium for electrodeposition of metals and alloys, improving morphology control and reducing parasitic reactions.
Catalysis and Synthesis
- Provides a non‑volatile, thermally stable reaction medium for transition‑metal catalysis, including cross‑coupling, hydrogenation, and oxidation systems.
- Supports biphasic catalysis and catalyst recycling due to its immiscibility with many organic solvents.
- Enhances selectivity and rate in ionic‑liquid‑assisted organic transformations where strong anion stability is beneficial.
Materials Science and Surface Engineering
- Used in polymer electrolyte formulations to improve ionic conductivity and mechanical stability in high‑performance membranes.
- Acts as a templating or structuring agent in the synthesis of porous materials, nanocomposites, and functional coatings.
- Serves as a lubricant or anti‑wear additive in high‑temperature or vacuum environments due to its low vapor pressure and thermal resilience.
Separation Science and Extraction
- Applied in liquid–liquid extraction systems for selective separation of metal ions, hydrophobic organics, and fluorinated species.
- Supports chromatographic method development as a stationary‑phase modifier or mobile‑phase additive, improving peak shape and selectivity.
Spectroscopy and Fundamental Research
- Provides a controlled, non‑volatile medium for spectroscopic studies (NMR, IR, UV‑Vis) of ionic transport, solvation, and ion‑pairing behavior.
- Used in fundamental investigations of pyridinium‑based ionic liquid structure–property relationships, especially those involving TFSI anion dynamics.
Please contact us if you want to learn more or need assistance with your order.