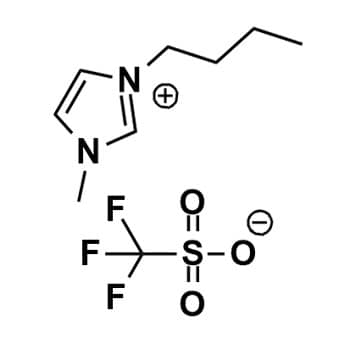
1-Butyl-3-methylimidazolium triflate, >99%
Price range: $114.58 through $6,501.15
Product Code: IL-0013-HPCAS NO: 174899-66-2
- Chemical Formula: C9H15F3N2O3S
- Synonym(s): BMIM Otf, C1C4Im OTf, Im 14 OTf, 1-Butyl-3-methylimidazolium trifluoromethanesulfonate
- Hydrophilic
- Aromatic
Conductivity: 3.05 mS/cm
SUM Formula: C9H15F3N2O3S
Molecular Weight: 288.29
Melting Point: 16 °C
Density: 1.30 g/cm³
Purity: >99%
Viscosity: 80.0 cP
- SUM Formula: C9H15F3N2O3S
- Molecular Weight: 288.29
- Melting Point: 16 °C
- Density: 1.30 g/cm³
- Viscosity: 80.0 cP
1-Butyl-3-methylimidazolium triflate, CAS: 174899-66-2
Key Applications:
- Gas Solubility & Greenhouse Gas Capture
- BMIM OTf is widely used in high‑pressure solubility studies for CO₂, N₂O, and CH₄, supporting research into gas separation, acid‑gas removal, and next‑generation carbon‑capture technologies. Its strong affinity for CO₂ makes it a valuable benchmark ionic liquid for evaluating gas–liquid equilibria and absorption mechanisms.
- Electrolytes for Fuel Cells & Electrochemical Devices
- This ionic liquid functions as a high‑stability electrolyte or electrolyte additive in proton‑exchange membrane fuel cells (PEMFCs) and high‑temperature polymer systems. Composite membranes incorporating BMIM OTf (e.g., SPEEK–BMIM OTf) demonstrate improved ionic conductivity, thermal stability, and mechanical performance.
- Polymer & Composite Material Engineering
- BMIM OTf is incorporated into polymer matrices to tune microstructure, enhance ionic transport, and improve thermal behavior. It is used in the development of ionic‑liquid‑based composite membranes for electrochemical and separation applications.
- Supported Ionic Liquid Phases (SILPs)
- BMIM‑based ionic liquids, including BMIM OTf, are immobilized on porous supports and metal–organic frameworks (MOFs) to create SILP materials with enhanced adsorption, catalytic activity, and gas‑uptake performance.
- Thermal Stability & Structure–Property Studies
- BMIM OTf is frequently used in fundamental research examining:
-
-
- Thermal decomposition pathways
- Cation–anion interactions
- Structure–property relationships in imidazolium ionic liquids
-
Please contact us if you want to learn more or need assistance with your order.