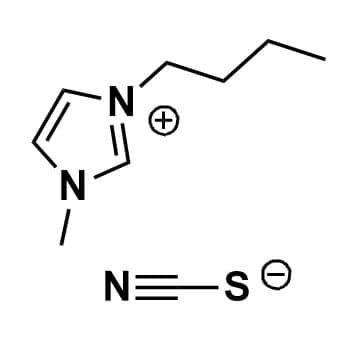
1-Butyl-3-methylimidazolium thiocyanate, >98%
Price range: $83.43 through $3,497.54
Product Code: IL-0063-HPCAS NO: 344790-87-0
- Chemical Formula: C9H15N3S
- Synonyms: BMIM SCN, C1C4Im SCN,Im 14 SCN
- Aromatic
Conductivity: 8.98 mS/cm (30 °C)
SUM Formula: C9H15N3S
Molecular Weight: 197.30
Melting Point: -47 °C
Density: 1.07 g/cm³ (30 °C)
Purity: >98%
Viscosity: 35.9 cP (25 °C)
- SUM Formula: C9H15N3S
- Molecular Weight: 197.30
- Melting Point: -47 °C
- Density: 1.07 g/cm³ (30 °C)
- Viscosity: 35.9 cP (25 °C)
1-Butyl-3-methylimidazolium thiocyanate, CAS: 344790-87-0
Key Applications:
Catalysis and Synthesis
- Serves as a polar, non‑volatile reaction medium for transition‑metal‑catalyzed transformations, including C–C and C–N bond‑forming reactions.
- Enhances rates and selectivity in nucleophilic substitution and heterocycle‑forming reactions due to the coordinating SCN⁻ anion.
- Supports biphasic and recyclable catalytic systems, reducing solvent waste and improving process efficiency.
Materials Processing and Polymer Science
- Effective solvent or co‑solvent for biopolymers such as cellulose, chitin, and lignin, enabling dissolution, regeneration, and functionalization workflows.
- Facilitates polymer blending, derivatization, and nanoparticle dispersion where strong anion coordination improves solubility and uniformity.
- Useful in ionothermal synthesis of porous materials, including metal–organic frameworks and inorganic nanostructures.
Electrochemistry and Energy Storage
- Functions as an electrolyte component in electrochemical devices, benefiting from the SCN⁻ anion’s conductivity and coordination properties.
- Supports redox‑active species in dye‑sensitized solar cells, electrochemical sensors, and electrodeposition systems.
- Enables stable ionic environments for studying charge‑transfer mechanisms and electrode interfaces.
Extraction, Separation, and Analytical Chemistry
- Applied in liquid–liquid extraction of metal ions, particularly soft or borderline metals that interact strongly with thiocyanate.
- Enhances selectivity in separation processes involving transition metals, rare‑earth elements, and organometallic complexes.
- Used as a medium for spectroscopic and electroanalytical studies where controlled solvation and low volatility are required.
Thermal and Process Engineering
- Suitable as a heat‑transfer or thermal‑stabilizing medium in specialized systems due to its low vapor pressure and thermal robustness.
- Supports high‑temperature reaction environments where conventional solvents degrade or volatilize.
Please contact us if you want to learn more or need assistance with your order.