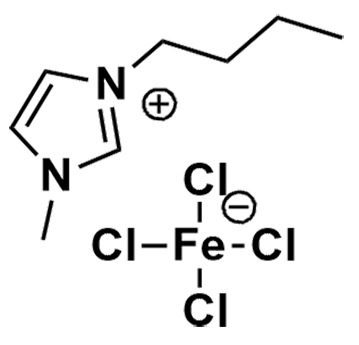
1-Butyl-3-methylimidazolium tetrachloroferrate(III), >97%
Price range: $84.92 through $3,221.66
Product Code: IL-0047-SGCAS NO: 359845-21-9
- Chemical Formula: C8H15Cl4FeN2
- Synonyms:BMIM FeCl4, C1C4in FeCl4, in14 FeCl4
- Ferromagnetic ionic liquid
- Aromatic
Conductivity: 4.37 mS/cm (25 °C)
SUM Formula: C8H15Cl4FeN2
Molecular Weight: 336.87
Melting Point: -7 °C
Density: 1.37 g/cm³ (25 °C)
Purity: >97%
Viscosity: 43.4 cP (25 °C)
- SUM Formula: C8H15Cl4FeN2
- Molecular Weight: 336.87
- Melting Point: -7 °C
- Density: 1.37 g/cm³ (25 °C)
- Viscosity: 43.4 cP (25 °C)
1-Butyl-3-methylimidazolium tetrachloroferrate(III), CAS: 359845-21-9
Key Applications:
Magnetically Responsive Solvents and Separation Media
- Functions as a magnetic ionic liquid (MIL) with tunable magnetic susceptibility, enabling remote, non‑contact manipulation of the solvent phase.
- Applied in liquid–liquid extraction systems where magnetic fields facilitate rapid phase disengagement and solvent recovery.
- Used in selective metal ion extraction, particularly for Fe‑based, lanthanide, and transition‑metal separations in hydrometallurgical workflows.
Catalysis and Reaction Media
- Serves as a Lewis acidic catalytic medium, supporting transformations such as Friedel–Crafts alkylation, esterification, and polymerization.
- Provides a dual‑function environment (ionic + magnetic) that enhances catalyst recyclability through magnetic retrieval.
- Employed as a task‑specific solvent for reactions requiring strong coordination to substrates or intermediates.
Electrochemical and Magnetic Materials Research
- Investigated as a component in magneto‑electrochemical systems, where magnetic fields modulate ionic transport or electrode processes.
- Used in the development of magnetically switchable electrolytes, enabling field‑controlled conductivity or viscosity.
- Supports fabrication of smart fluids and stimuli‑responsive materials, including magneto‑rheological formulations.
Environmental and Analytical Applications
- Enables magnetically assisted microextraction (MA‑ME) techniques for trace analyte enrichment in environmental and biological samples.
- Utilized in solvent‑free or reduced‑solvent analytical workflows, where magnetic recovery minimizes waste and simplifies cleanup.
Nanomaterials and Hybrid Systems
- Acts as a stabilizing and dispersing medium for magnetic nanoparticles, improving colloidal stability and enabling field‑guided assembly.
- Supports synthesis of ionic‑liquid‑coated magnetic nanocomposites for catalysis, sensing, and adsorption.
Please contact us if you want to learn more or need assistance with your order.