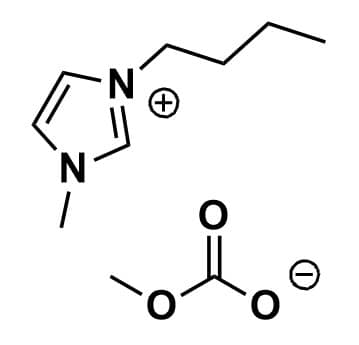
1-Butyl-3-methylimidazolium methylcarbonate, >97%, 30% in MeOH
Price range: $42.39 through $440.66
Product Code: IN-0028-SGCAS NO: 916850-37-8
- Chemical Formula: C10H18N2O3
- Synonyms: BMIM MeCO3, C1C4Im MeCO3, Im14 MeCO3
- Hydrophilic
To order 5kg or more, please email us at info@roco.global
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C10H18N2O3
Molecular Weight: 214.13
Purity: >97%
- SUM Formula: C10H18N2O3
- Molecular Weight: 214.13
1-Butyl-3-methylimidazolium methylcarbonate, CAS: 916850-37-8
Key Applications:
Solvent and Medium for Organic Synthesis
- Functions as a polar, non‑volatile reaction medium for nucleophilic substitutions, condensations, and base‑sensitive transformations.
- Supports homogeneous catalysis and transition‑metal–mediated coupling reactions by stabilizing reactive intermediates.
- The methylcarbonate anion provides mild basicity, enabling selective deprotonation without the harshness of inorganic bases.
CO₂‑Responsive and Carbonate‑Based Chemistry
- Useful in CO₂‑capture and CO₂‑activation studies due to the reversible nature of the methylcarbonate anion.
- Serves as a model ionic liquid for probing carbonate equilibria, anion exchange, and CO₂‑triggered speciation changes.
- Employed in developing reversible CO₂‑binding solvents and switchable polarity systems.
Polymer Processing and Functional Material Development
- Acts as a processing aid for cellulose, chitosan, and other biopolymers that require strong hydrogen‑bond disruption.
- Supports the preparation of carbonate‑functionalized polymers and polymer electrolytes.
- Enables dissolution and regeneration workflows for sustainable materials research.
Electrochemical and Energy‑Related Applications
- Provides a stable ionic environment for electrochemical studies, including electrodeposition and redox‑active species stabilization.
- Investigated as a component in electrolyte formulations for low‑volatility, non‑flammable systems.
- The carbonate anion contributes to SEI‑relevant decomposition pathways, making it useful in mechanistic battery research.
Catalysis and Reaction Engineering
- Enhances catalytic turnover in base‑assisted reactions due to its intrinsic anion basicity.
- Supports organocatalytic transformations where carbonate species participate in proton shuttling or transient activation.
- Compatible with biphasic catalysis and catalyst recycling strategies.
Separation Science and Extraction
- Applied in liquid–liquid extraction of metal ions, organic acids, and polar organics.
- The carbonate anion modulates extraction selectivity through tunable hydrogen‑bonding and acid–base interactions.
- Useful in designing greener extraction systems with reduced volatility and improved recyclability.
Analytical and Spectroscopic Studies
- Serves as a reference ionic liquid for studying carbonate speciation, ion pairing, and solvent–solute interactions.
- Suitable for NMR, IR, and conductivity studies due to its well‑defined structure and high purity.
- The methanol solution format (30% in MeOH) improves handling and facilitates direct injection into analytical workflows.
Please Contact Us if you want to learn more or need assistance with your order.