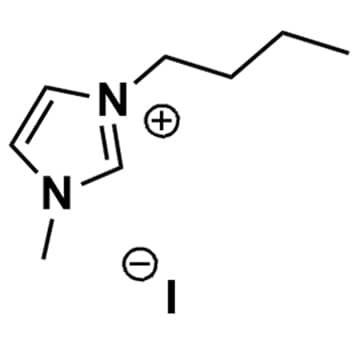
1-Butyl-3-methylimidazolium iodide, >98%
Price range: $143.02 through $1,535.73
Product Code: IL-0051-HPCAS NO: 65039-05-6
- Chemical Formula: C8H15IN2
- Synonyms: BMIM I, C1C4Im I,Im 14 I
- Light Sensitive
- Hydrophilic
Conductivity: 0.52 mS/cm
SUM Formula: C8H15IN2
Molecular Weight: 266.12
Melting Point: -72 °C
Density: 1.48 g/cm³
Purity: >98%
Viscosity: 1183 cP
- SUM Formula: C8H15IN2
- Molecular Weight: 266.12
- Melting Point: -72 °C
- Density: 1.48 g/cm³
- Viscosity: 1183 cP
1-Butyl-3-methylimidazolium iodide, CAS: 65039-05-6
Key Applications:
Electrochemical Systems & Redox Chemistry
- Used as a supporting electrolyte and ionic conductor in electrochemical cells, benefiting from its high ionic mobility and thermal stability.
- Employed in redox‑active systems, including iodide/tri‑iodide couples, due to the stabilizing effect of the imidazolium cation on halide species.
Dye‑Sensitized Solar Cells (DSSCs)
- Functions as a key iodide source in liquid electrolytes for DSSCs, enabling efficient charge transport and regeneration of the oxidized dye.
- Enhances device stability through strong ion pairing and low volatility.
Catalysis & Organic Synthesis
- Serves as a halide‑rich ionic medium for nucleophilic substitution, phase‑transfer catalysis, and metal‑catalyzed transformations.
- Provides a tunable, non‑volatile reaction environment that improves selectivity and yields in halide‑dependent reactions.
Solvent & Reaction Medium
- Acts as a polar, aprotic ionic liquid solvent for reactions requiring strong halide presence, including SN1/SN2 pathways and transition‑metal catalysis.
- Supports high‑temperature and microwave‑assisted synthesis due to its thermal robustness.
Materials Processing & Nanomaterials
- Utilized in the synthesis and stabilization of metal nanoparticles, where iodide ions influence nucleation and growth behavior.
- Applied in templating and surface‑modification processes for carbon materials, polymers, and inorganic frameworks.
Electrolytes for Batteries & Energy Storage
- Investigated as a component in electrolytes for next‑generation batteries, where iodide anions contribute to redox‑mediated charge storage mechanisms.
- Offers high ionic conductivity and wide electrochemical windows suitable for experimental energy‑storage architectures.
Biomass Conversion & Green Chemistry
- Provides a halide‑rich ionic environment for depolymerization of lignocellulosic biomass and activation of carbohydrate substrates.
- Supports catalytic upgrading pathways in sustainable chemical processing.
Please contact us if you want to learn more or need assistance with your order.