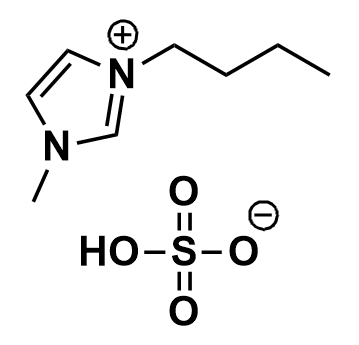
1-Butyl-3-methylimidazolium hydrogensulfate, >98%
Price range: $130.16 through $1,453.95
Product Code: IL-0060-HPCAS NO: 262297-13-2
- Chemical Formula: C8H16N2O4S
- Synonyms: 1-butyl-3-methylimidazolium bisulfate, BMIM HSO4, C1C4in HSO4,14 HSO4
- Hydrophilic
- Aromatic
** This product will incur a $97.00 HazMat fee when the order is placed. **
Conductivity: 0.10 mS/cm
SUM Formula: C8H16N2O4S
Molecular Weight: 236.29
Melting Point: 33 °C
Density: 1.28 g/cm³
ECW: 3.0 V
Purity: >98%
Viscosity: 3088 cP
- SUM Formula: C8H16N2O4S
- Molecular Weight: 236.29
- Melting Point: 33 °C
- Density: 1.28 g/cm³
- ECW: 3.0 V
- Viscosity: 3088 cP
1-Butyl-3-methylimidazolium hydrogensulfate, CAS: 262297-13-2
Key Applications:
Catalysis and Organic Synthesis
- Functions as a strong, thermally stable Brønsted acidic ionic liquid for esterification, transesterification, alkylation, and dehydration reactions.
- Enables solvent–catalyst dual functionality, reducing the need for mineral acids in biomass conversion and fine‑chemical synthesis.
- Supports acid‑catalyzed transformations under mild conditions with improved selectivity and recyclability.
Biomass Processing and Green Chemistry
- Used in hydrolysis and depolymerization of lignocellulosic biomass due to its acidity and ability to disrupt hydrogen‑bond networks.
- Facilitates conversion of carbohydrates into platform chemicals such as HMF and levulinic acid.
- Aligns with sustainable‑processing goals by reducing corrosive waste streams and enabling closed‑loop solvent systems.
Electrochemical and Energy Applications
- Serves as a proton‑conducting ionic liquid for electrochemical devices requiring non‑volatile, thermally stable electrolytes.
- Investigated in fuel‑cell membranes and proton‑exchange systems where water‑free proton transport is advantageous.
- Provides high ionic conductivity and chemical stability across broad temperature ranges.
Separation Science and Extraction
- Acts as a tunable, acidic extraction medium for metal ions, dyes, and organic compounds.
- Useful in biphasic catalytic systems and liquid–liquid extraction workflows where acidity enhances partitioning behavior.
- Supports selective dissolution of inorganic salts and metal oxides.
Materials Processing
- Employed as a processing aid for cellulose, chitin, and other hydrogen‑bonded polymers.
- Enhances dissolution and regeneration processes for advanced materials and membranes.
- Used in nanoparticle synthesis where acidic ionic environments control nucleation and growth.
Industrial and Process Advantages
- High thermal stability and negligible vapor pressure improve safety and reduce emissions.
- Recyclable across multiple reaction cycles, lowering operational costs.
- Compatible with continuous‑flow systems and high‑throughput catalytic platforms.
Please contact us if you want to learn more or need assistance with your order.