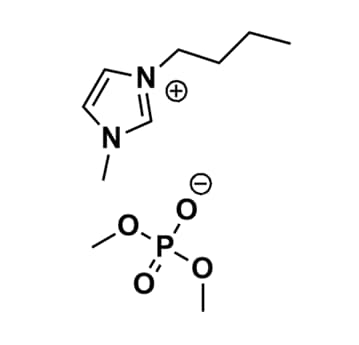
1-Butyl-3-methylimidazolium dimethyl phosphate, >98%
Price range: $271.69 through $2,468.49
Product Code: IL-0257-HPCAS NO: 891772-94-4
- Chemical Formula: C10H21N2O4P
- Synonyms: BMIM DEP, C1C4Im DEP, Im14 DEP
- Aromatic
Conductivity: 0.27 mS/cm (30 °C)
SUM Formula: C10H21N2O4P
Molecular Weight: 264.26
Melting Point: -28 °C
Density: 1.16 g/cm³ (20 °C)
Purity: >98%
Viscosity: 534 cP (25 °C)
- SUM Formula: C10H21N2O4P
- Molecular Weight: 264.26
- Melting Point: -28 °C
- Density: 1.16 g/cm³ (20 °C)
- Viscosity: 534 cP (25 °C)
1-Butyl-3-methylimidazolium dimethyl phosphate, CAS: 891772-94-4
Key Applications:
Solvent and Processing Applications
- Biomass dissolution and fractionation
Effective at solubilizing lignocellulosic materials, enabling pretreatment, delignification, and selective extraction of cellulose or hemicellulose for biofuel and biopolymer workflows.
- Catalyst and reagent medium
Supports acid‑catalyzed and base‑catalyzed transformations due to its relatively polar, hydrogen‑bond‑accepting phosphate anion. Used in esterification, transesterification, and dehydration reactions where conventional solvents underperform.
- Polymer processing and modification
Facilitates dissolution or swelling of biopolymers such as cellulose, chitosan, and starch. Useful for membrane casting, fiber spinning, and polymer blending under mild conditions.
Electrochemical and Energy Applications
- Electrolyte component for moderate‑temperature systems
The dimethyl phosphate anion provides good ionic conductivity and moderate viscosity, making it suitable for exploratory electrolyte formulations in supercapacitors and electrochemical cells.
- Ion‑transport medium in redox systems
Supports stable ion mobility and can be blended with other ionic liquids or organic carbonates to tune conductivity and viscosity.
Extraction and Separation
- Selective extraction of metal ions
Demonstrates affinity for certain transition‑metal species, enabling use in liquid–liquid extraction, hydrometallurgical separations, and recovery of critical metals.
- Organic compound separation
Useful in partitioning phenolics, organic acids, and other polar organics due to its tunable polarity and low volatility.
Catalysis and Green Chemistry
- Reaction medium for sustainable synthesis
Non‑volatile and thermally stable, enabling recyclable solvent systems for acid/base catalysis, biocatalysis, and organometallic transformations.
- Support for immobilized catalysts
Provides a stable ionic environment for enzymes, nanoparticles, and homogeneous catalysts, improving turnover and reusability.
Materials and Surface Science
- Precursor or additive in functional coatings
Enhances ionic conductivity and hydrophilicity in polymer coatings, gels, and thin films.
- Template or structuring agent
Assists in forming ordered inorganic or hybrid materials, including mesoporous oxides and phosphates.
Thermal and Lubrication Uses
- High‑temperature lubricant additive
The phosphate anion contributes anti‑wear and anti‑scuff properties, making it suitable for exploratory lubrication formulations.
- Heat‑transfer medium
Low vapor pressure and thermal stability support use in controlled‑temperature environments where conventional organics degrade.
Please contact us if you want to learn more or need assistance with your order.