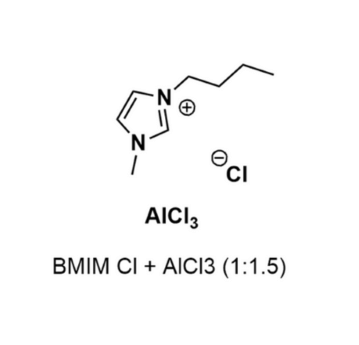
1-Butyl-3-methylimidazolium chloride and Aluminum chloride (1:1.5)
Price range: $86.41 through $584.62
Product Code: EP-0002-HPCAS NO: 80432-09-3
- Chemical Formula: C₈H₁₅N₂]Cl·1.5AlCl₃
- Synonyms: BMIM Cl/AlCl3 (1:1,5), BMIM AlCl4
- Electrolyte for aluminum deposition
*This product will incur a $97.00 HazMat fee when the order is placed.*
SUM Formula: C₈H₁₅N₂]Cl·1.5AlCl₃
Purity: NA
- SUM Formula: C₈H₁₅N₂]Cl·1.5AlCl₃
1-Butyl-3-methylimidazolium chloride and Aluminum chloride (1:1.5), CAS: 80432-09-3
Key Applications:
Lewis‑Acidic Catalysis
- Serves as a strongly Lewis‑acidic ionic liquid for Friedel–Crafts alkylation and acylation.
- Enables high‑selectivity transformations in isomerization, oligomerization, and alkylation of aromatics.
- Functions as a reusable catalytic medium, reducing reliance on volatile organic solvents.
Electrochemical Systems
- Used as an electrolyte in aluminum electrodeposition, enabling smooth, dense aluminum coatings at moderate temperatures.
- Supports electrochemical synthesis routes requiring moisture‑sensitive, chloride‑rich environments.
- Provides wide electrochemical windows suitable for metal plating and corrosion‑resistant surface finishing.
Metal Processing and Refining
- Applied in aluminum recovery and purification processes due to its ability to solubilize aluminum species.
- Facilitates controlled deposition of aluminum alloys for microelectronics and protective coatings.
Organic Synthesis and Reaction Media
- Acts as a dual‑function solvent–catalyst for chlorination, polymerization, and cationic rearrangements.
- Stabilizes reactive carbocations and metal complexes, enabling transformations not feasible in conventional solvents.
- Supports biphasic catalysis where product separation is simplified by the ionic liquid phase.
Materials Science and Thin‑Film Fabrication
- Used in the preparation of aluminum‑based thin films for microfabrication and semiconductor research.
- Provides a tunable medium for templated nanostructure formation and controlled metal nucleation.
Green Chemistry and Process Intensification
-
- Reduces hazardous waste by replacing corrosive mineral acids in Lewis‑acid catalysis.
- Offers recyclability and thermal stability, supporting closed‑loop reaction systems.
Please contact us if you want to learn more or need assistance with your order.
- Nahian, Md Khalid, and Ramana G. Reddy. “Electrical conductivity and species distribution of aluminum chloride and 1‐butyl‐3‐methylimidazolium chloride ionic liquid electrolytes.” Journal of Physical Organic Chemistry 36.10 (2023): e4549.
- Lang, Haiyan, et al. “Template-free preparation of spherical Al particles in aluminum chloride and 1-butyl-3-methylimidazolium chloride ionic liquid.” Ionics 24 (2018): 1781-1788.