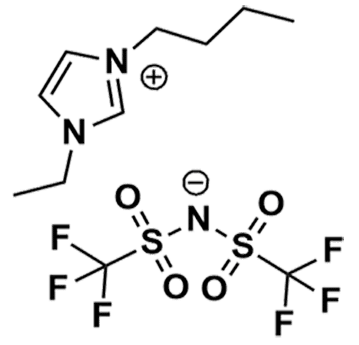
1-Butyl-3-ethylimidazolium bis(trifluoromethylsulfonyl)imide, 98%
Price range: $148.58 through $7,547.27
Product Code: IL-0289-HPCAS NO: 174899-89-9
- Chemical Formula: C11H17F6N3O4S2
- Synonyms: AT BTA, AT NTf 2, AT TFSI, AT BTI, C 2 C 4 Im BTA, Im 24 BTA
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
SUM Formula: C11H17F6N3O4S2
Molecular Weight: 433.39
Melting Point: <RT
Purity: >98%
- SUM Formula: C11H17F6N3O4S2
- Molecular Weight: 433.39
- Melting Point:
1-Butyl-3-ethylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 174899-89-9
Key Applications
Electrochemical Systems
- Serves as a high‑conductivity ionic liquid electrolyte for lithium, sodium, and multivalent metal batteries.
- Used in supercapacitors and hybrid capacitors where low viscosity and wide electrochemical stability windows are required.
- Supports electrodeposition of reactive or air‑sensitive metals under moisture‑free, non‑volatile conditions.
- Enables stable operation in high‑temperature electrochemical devices due to the thermal robustness of the BETI anion.
Catalysis and Synthesis
- Functions as a non‑volatile reaction medium for transition‑metal catalysis, including cross‑coupling, hydrogenation, and carbon–carbon bond‑forming reactions.
- Provides enhanced solubility for organometallic complexes, improving turnover and catalyst lifetime.
- Used in biphasic catalysis where phase separation simplifies product isolation.
- Supports acid‑sensitive transformations due to the weakly coordinating nature of the bis(trifluoromethylsulfonyl)imide anion.
Separation Science and Extraction
- Applied in liquid–liquid extraction of metal ions, particularly rare‑earth and actinide species, where hydrophobicity and tunable coordination behavior improve selectivity.
- Used in extractive desulfurization and denitrification of fuels.
- Serves as a stationary‑phase component or additive in chromatography to modulate retention and selectivity.
Materials Processing
- Acts as a plasticizer and ion‑conducting additive in polymer electrolytes and ionogels.
- Supports fabrication of thin films, coatings, and membranes requiring low volatility and controlled ionic mobility.
- Used in nanoparticle synthesis as a stabilizing, non‑volatile medium that enables narrow size distributions.
Thermal and Chemical Stability Applications
- Suitable for high‑temperature reaction environments due to the thermal stability of the BETI anion.
- Employed in systems requiring low vapor pressure and chemical inertness, including vacuum‑compatible processes.
Spectroscopy and Analytical Chemistry
- Provides a low‑volatility matrix for mass spectrometry and spectroscopic studies of ionic species.
- Used as a solvent for electroanalytical measurements where wide potential windows are essential.
Please contact us if you want to learn more or need assistance with your order.