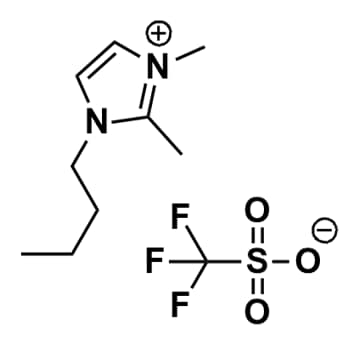
1-Butyl-2,3-dimethylimidazolium triflate, >98%
Price range: $278.87 through $3,848.60
Product Code: IL-0059-HPCAS NO: 765910-73-4
- Chemical Formula: C10H17F3N2O3S
- Synonyms: BDiMIM OTf, Im114 OTf,1-butyl-2,3-dimethylimidazolium trifluoromethanesulfonate
- Hydrophobic
- Aromatic
SUM Formula: C10H17F3N2O3S
Molecular Weight: 302.32
Melting Point: 40 °C
Purity: >98%
- SUM Formula: C10H17F3N2O3S
- Molecular Weight: 302.32
- Melting Point: 40 °C
1-Butyl-2,3-dimethylimidazolium triflate, CAS: 765910-73-4
Key Applications:
1. Catalysis and Organic Synthesis
- Brønsted‑acidic reaction media for esterifications, acylations, alkylations, and dehydrations.
- Solvent–catalyst hybrid for Friedel–Crafts reactions, Diels–Alder cycloadditions, and electrophilic aromatic substitutions.
- Stabilizing medium for carbocationic and metal‑centered catalytic cycles, due to the triflate anion’s weak coordination.
- Recyclable reaction solvent, reducing solvent waste in multi‑step synthesis.
2. Electrochemistry and Energy Materials
- Electrolyte component in supercapacitors and electrochemical capacitors where high ionic conductivity and wide electrochemical windows are required.
- Solvent for redox‑active species in flow‑battery research.
- Medium for electrodeposition of metals or alloys where moisture‑sensitive or air‑sensitive conditions are needed.
3. Separation Science and Extraction
- Selective extraction of metal ions, especially where hydrophobic ionic liquids improve phase separation.
- Solvent for biomass fractionation, including lignin dissolution and carbohydrate processing.
- CO₂ and SO₂ capture studies, leveraging the imidazolium cation’s tunable interactions with acidic gases.
4. Materials Processing
- Template or structuring agent in polymer synthesis, block‑copolymer self‑assembly, and nanoparticle stabilization.
- Plasticizer or ionic additive in polymer electrolytes and ion‑conducting films.
- Medium for cellulose dissolution and regeneration, depending on the specific hydrogen‑bonding profile of the triflate system.
5. Spectroscopy and Physical Chemistry
- Non‑volatile matrix for studying solvation dynamics, ion pairing, and transport properties.
- Model system for benchmarking viscosity, conductivity, and thermal stability trends across substituted imidazolium ionic liquids.
6. Green Chemistry and Process Intensification
- Replacement for volatile organic solvents in reactions requiring high temperature or strong acidity.
- Recyclable reaction medium in continuous‑flow or biphasic catalytic systems.
- Platform for immobilizing homogeneous catalysts, improving catalyst lifetime and recovery.
Please contact us if you want to learn more or need assistance with your order.