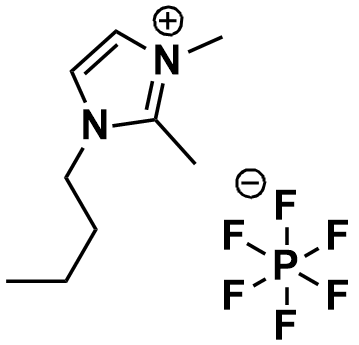
1-Butyl-2,3-dimethylimidazolium hexafluorophosphate, >99%
Price range: $142.89 through $8,208.21
Product Code: IL-0057-HPCAS NO: 227617-70-1
- Chemical Formula: C9H17F6N2P
- Synonyms: BDiMIM PF6, IM114 PF6
- Anion decomposes slowly in the presence of water
- Weakly coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 0.76 mS/cm (45 °C)
SUM Formula: C9H17F6N2P
Molecular Weight: 298.21
Melting Point: 43 °C
Density: 1.444 g/cm³
Purity: >99%
Viscosity: 554 cP (45 °C)
- SUM Formula: C9H17F6N2P
- Molecular Weight: 298.21
- Melting Point: 43 °C
- Density: 1.444 g/cm³
- Viscosity: 554 cP (45 °C)
1-Butyl-2,3-dimethylimidazolium hexafluorophosphate, CAS: 227617-70-1
Key Applications:
- Electrochemical Systems & Ionic‑Liquid Electrolytes
-
- Serves as a hydrophobic, wide‑electrochemical‑window electrolyte for supercapacitors, dye‑sensitized solar cells (DSSCs), and redox‑flow systems.
- PF₆⁻ anion provides low viscosity relative to TFSI analogs, enabling improved ion mobility in mixed‑solvent systems.
- The 2,3‑dimethyl substitution increases cation symmetry, reducing rotational barriers and enhancing thermal and electrochemical stability.
- Solvent for Organometallic & Transition‑Metal Catalysis
-
- Effective medium for C–C coupling, hydrogenation, and oxidation reactions due to high polarity and low coordinating ability.
- Hydrophobicity allows biphasic catalysis with easy product separation and catalyst recycling.
- Extraction & Separation Science
-
- Useful in liquid–liquid extraction of metal ions (e.g., rare earths, transition metals) due to strong PF₆⁻ hydrophobicity.
- Demonstrates tunable partitioning behavior for aromatic hydrocarbons, phenolics, and bio‑derived molecules.
- Thermal Fluids & Heat‑Transfer Media
-
- High thermal stability and negligible vapor pressure make it suitable for heat‑transfer fluids, thermostats, and high‑temperature reaction media.
- The 2,3‑dimethyl pattern reduces crystallinity, improving low‑temperature fluidity.
- Lubrication & Tribology
-
- Ionic liquids with PF₆⁻ anions form robust boundary films on steel and ceramic surfaces.
- Demonstrated reductions in friction coefficients and wear rates under high load and temperature.
- Materials Processing & Nanomaterials Synthesis
-
- Acts as a templating medium for nanoparticle synthesis (Au, Pt, Fe₃O₄).
- Supports formation of nanostructured carbons, polymer composites, and ionic‑liquid‑modified membranes.
Please contact us if you want to learn more or if you need assistance with your order.