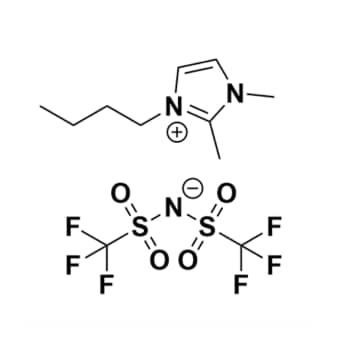
1-Butyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $96.16 through $6,366.66
Product Code: IL-0104-HPCAS NO: 350493-08-2
- Chemical Formula: C11H17F6N3O4S2
- SYNONYMS: BMMIm NTf2
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 1.96 mS/cm (25 °C)
SUM Formula: C11H17F6 N3O4S2
Molecular Weight: 433.39
Melting Point: -44 °C
Density: 1.42 g/cm³
ECW: 4.6 V
Purity: >99%
Viscosity: 98.0 cP
- SUM Formula: C11H17F6 N3O4S2
- Molecular Weight: 433.39
- Melting Point: -44 °C
- Density: 1.42 g/cm³
- ECW: 4.6 V
- Viscosity: 98.0 cP
1-Butyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)imide, CAS NO: 350493-08-2
Key Applications:
Electrochemical Applications
- High‑voltage electrolytes for lithium, sodium, and multivalent metal batteries, leveraging wide electrochemical windows and low flammability.
- Electrolyte additive or co‑solvent to improve SEI formation, ionic conductivity, and thermal stability in next‑generation cell chemistries.
- Supercapacitor electrolytes, particularly for high‑temperature or high‑power systems requiring low volatility and stable ion transport.
- Electrodeposition media for metals and alloys where controlled nucleation and smooth film formation are required.
Catalysis and Synthesis
- Non‑coordinating reaction medium for acid‑sensitive or moisture‑sensitive transformations due to the weakly coordinating TFSI⁻ anion.
- Phase‑transfer medium enabling enhanced solubility of organic substrates and transition‑metal complexes.
- Support for homogeneous and biphasic catalysis, especially in C–C coupling, hydrogenation, and oxidation systems.
- Stabilizing environment for reactive intermediates, benefiting mechanistic studies and high‑selectivity transformations.
Materials Science and Engineering
- Polymer electrolyte component for ion‑conducting membranes, gels, and solid‑state devices requiring high thermal stability.
- Plasticizer or ionic dopant in advanced polymers, improving conductivity and mechanical flexibility.
- Processing aid for nanomaterials, including dispersion of carbon nanotubes, graphene, and metal nanoparticles.
- Template or structuring medium in sol–gel and porous material synthesis where ionic liquids influence pore architecture.
Separation and Extraction
- Hydrophobic extraction phase for metal ions, organometallic species, and hydrophobic organics.
- Solvent for liquid–liquid extraction in systems requiring low miscibility with water and high chemical stability.
- Selective dissolution medium for fluorinated compounds, aromatic species, and specialty polymers.
Thermal and Chemical Stability Applications
- Heat‑transfer and thermal‑management fluids in niche systems requiring non‑volatile, thermally stable media.
- Lubrication and anti‑wear additives in extreme environments where conventional lubricants degrade.
- Stabilizing medium for high‑temperature spectroscopy, electrochemical testing, and reaction monitoring.
Analytical and Research Uses
- Electrochemical probe solvent for studying redox couples, ion pairing, and charge‑transfer kinetics.
- Medium for spectroscopic studies (NMR, IR, UV‑Vis) where low vapor pressure and chemical inertness are advantageous.
- Reference formulation component in benchmarking ionic liquid behavior across cation/anion families.
Please contact us if you want to learn more or need assistance with your order.
You may also like…
-
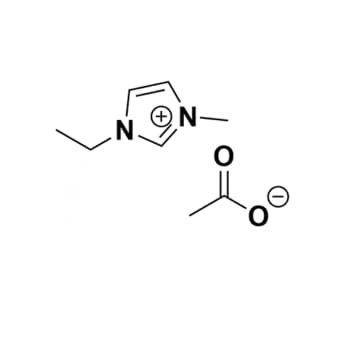
1-Ethyl-3-methylimidazolium acetate, >95%
Price range: $41.04 through $2,378.02Product Code: IL-0189-TG -
1-Butyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $96.16 through $6,366.66Product Code: IL-0104-HP -
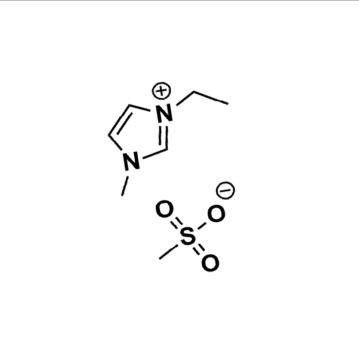
1-Ethyl-3-methylimidazolium methanesulfonate, >99%
Price range: $97.65 through $2,477.02Product Code: IL-0004-HP