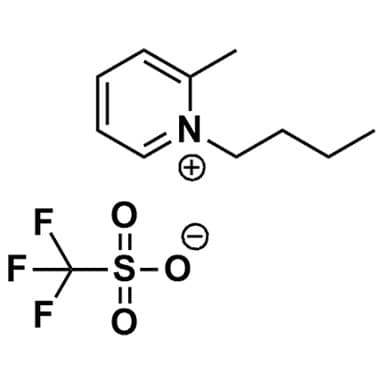
1-Butyl-2-methylpyridinium triflate, >99%
Price range: $628.43 through $5,033.27
Product Code: IL-0178-HPCAS NO: 1770850-20-8
- Chemical Formula: C11H16F3NO3S
- Synonyms: Bu-2-Pic OTf, Bu2Pic OTf, 1-Butyl-2-methylpyridinium trifluoromethanesulfonate, 1-Butyl-2-picolinium triflate, N-Butyl-2-methylpyridinium triflate, N-Butyl-2-picolinium triflate
- Aromatic
Conductivity: 1.47 mS/cm (30 °C)
SUM Formula: C11H16F3NO3S
Molecular Weight: 299.31
Melting Point: <RT
Density: 1.30 g/cm³ (26 °C)
Purity: >98%
Viscosity: 240 cP (25°C)
- SUM Formula: C11H16F3NO3S
- Molecular Weight: 299.31
- Melting Point:
- Density: 1.30 g/cm³ (26 °C)
- Viscosity: 240 cP (25°C)
1-Butyl-2-methylpyridinium triflate, CAS: 1770850-20-8
Key Applications:
Electrochemical Systems
- Serves as a stable ionic liquid electrolyte component with wide electrochemical windows.
- Supports ion transport in supercapacitors, redox‑flow systems, and specialty battery chemistries.
- Useful in electrodeposition studies where low volatility and controlled ion mobility are required.
Catalysis and Synthesis
- Functions as a non‑volatile reaction medium for acid‑catalyzed and transition‑metal‑catalyzed transformations.
- Enhances selectivity in electrophilic and nucleophilic substitution reactions due to its weakly coordinating triflate anion.
- Provides a tunable solvent environment for organometallic catalysis and biphasic reaction systems.
Materials Science
- Employed in polymer electrolyte formulations to improve ionic conductivity and thermal stability.
- Acts as a processing aid for ion‑conductive films, membranes, and specialty coatings.
- Supports the fabrication of ionic‑liquid‑modified nanoparticles and hybrid materials.
Separation Science
- Used as a stationary‑phase modifier in chromatography to adjust polarity and retention behavior.
- Enables selective extraction of metal ions and organic species in liquid–liquid or supported liquid membrane systems.
Thermal and Transport Studies
- Suitable for fundamental investigations of viscosity, conductivity, and ion‑pairing behavior in pyridinium‑based ionic liquids.
- Provides a model system for studying structure–property relationships in substituted pyridinium salts.
Please contact us if you would like to learn more or need assistance with your order.