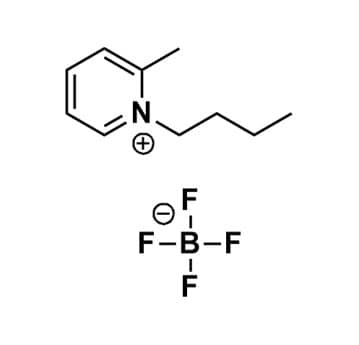
1-Butyl-2-methylpyridinium tetrafluoroborate, >99%
Price range: $224.96 through $9,837.39
Product Code: IL-0111-HPCAS NO: 286453-46-1
- Chemical Formula: C10H16BF4N
- Synonyms: Bu-2-Pic BF4, Bu2Pic BF4, 1-Butyl-2-picolinium tetrafluoroborate, N-Butyl-2-methylpyridinium tetrafluoroborate, N-Butyl-2-picolinium tetrafluoroborate
- Anion decomposes slowly in the presence of water.
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 1.83 mS/cm (23 °C)
SUM Formula: C10H16BF4N
Molecular Weight: 237.05
Melting Point: 12 °C
Density: 1.203 g/cm³ (25 °C)
Purity: >99%
Viscosity: 258 cP (20 °C)
- SUM Formula: C10H16BF4N
- Molecular Weight: 237.05
- Melting Point: 12 °C
- Density: 1.203 g/cm³ (25 °C)
- Viscosity: 258 cP (20 °C)
1-Butyl-2-methylpyridinium tetrafluoroborate, CAS: 286453-46-1
Key applications:
Electrochemical Systems
- Serves as a high‑conductivity electrolyte component for supercapacitors, redox‑flow systems, and advanced battery research.
- Supports stable ion transport under wide potential windows due to the BF₄⁻ anion’s electrochemical robustness.
- Useful as a co‑solvent or ionic additive to tune viscosity, conductivity, and interfacial behavior in electrode–electrolyte systems.
Catalysis and Synthesis
- Functions as a non‑volatile reaction medium for transition‑metal catalysis, enabling improved selectivity and recyclability.
- Provides a polar, tunable environment for nucleophilic substitutions, cyclizations, and heterocycle functionalization.
- Suitable for biphasic catalysis where phase separation and catalyst recovery are required.
Separation Science
- Acts as an extraction medium for metal ions, organics, and polar analytes, leveraging its cation–anion pairing to modulate solvation.
- Supports ionic‑liquid‑based liquid–liquid extraction systems where low vapor pressure and thermal stability are advantageous.
- Can be incorporated into supported ionic‑liquid membranes for selective transport of charged or polar species.
Materials and Surface Engineering
- Serves as a structuring agent or templating medium in the preparation of nanomaterials, conductive films, and porous frameworks.
- Useful in electrodeposition environments where ionic liquids improve morphology control and suppress side reactions.
- Provides a stable medium for polymerization processes requiring controlled ionic strength and low volatility.
Analytical and Spectroscopic Use
- Functions as a solvent for spectroscopic studies where minimal background volatility and broad thermal stability are required.
- Supports electroanalytical experiments by stabilizing reactive intermediates and enabling wide potential sweeps.
General Research Applications
- Suitable for fundamental studies of ion transport, solvation dynamics, and ionic‑liquid structure–property relationships.
- Provides a benchmark pyridinium‑based IL for comparative studies across BF₄⁻ systems.
Contact us if you want to learn more or need assistance with your order.