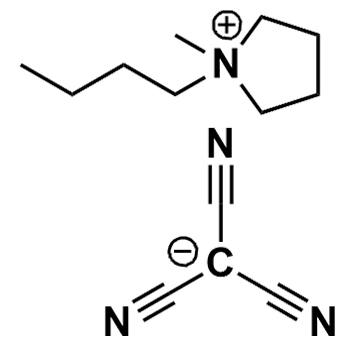
1-Butyl-1-methylpyrrolidinium tricyanomethanide, >98%
Price range: $151.42 through $1,632.03
Product Code: IL-0318-HPCAS NO: 878027-72-6
- Chemical Formula: C13H20N4
- Synonyms: BMPyrr TCM, PYR 14 TCM, PY 14 TCM, N-Butyl-N-methylpyrrolidinium tricyanomethanide
- Hydrophilic
Conductivity: 7.74 mS/cm (30 °C)
SUM Formula: C13H20N4
Molecular Weight: 232.32
Melting Point: <RT
Density: 1.01 g/ml (22°C)
ECW: 4.2V
Purity: >98%
Viscosity: 26.9 cP (25 °C)
- SUM Formula: C13H20N4
- Molecular Weight: 232.32
- Melting Point:
- Density: 1.01 g/ml (22°C)
- ECW: 4.2V
- Viscosity: 26.9 cP (25 °C)
1-Butyl-1-methylpyrrolidinium tricyanomethanide, CAS: 878027-72-6
Key Applications:
1. High‑Stability Ionic Liquid for Electrochemical Systems
- Serves as a thermally stable ionic liquid with a highly delocalized tricyanomethanide (TCM⁻) anion.
- Used in electrochemical cells, ionic liquid electrolytes, and redox‑active media where low viscosity and wide electrochemical windows are required.
- Supports research into next‑generation battery electrolytes, including organic redox systems and hybrid ion devices.
2. Conductive Media for Organic Electronics
- Employed as a tunable ionic conductor in organic field‑effect transistors (OFETs), electrochromic devices, and soft electronic interfaces.
- The TCM⁻ anion enhances charge mobility and reduces ion‑pairing, improving device responsiveness.
3. Solvent and Co‑solvent for Catalysis & Synthesis
- Functions as a polar, non‑volatile medium for transition‑metal catalysis, organocatalysis, and anion‑stabilized reaction pathways.
- The tricyanomethanide anion stabilizes reactive intermediates, enabling transformations that benefit from strong electron delocalization.
4. Template & Structure‑Directing Agent in Materials Chemistry
- Used in the preparation of nanoporous materials, coordination frameworks, and ionic‑liquid‑derived carbon materials.
- The TCM⁻ anion’s geometry and charge distribution influence pore formation and carbon microstructure during pyrolysis.
5. Thermal‑Stability Studies & Fundamental Ionic Liquid Research
- Serves as a model system for studying:
- anion delocalization effects
- ion‑pairing and transport
- structure–property relationships in pyrrolidinium ionic liquids
- Frequently used in comparative studies with BF₄⁻, PF₆⁻, TFSI⁻, and other anions.
6. Antistatic & Surface‑Modification Research
- Investigated as a component in antistatic coatings, surface‑charge dissipative layers, and ionic‑liquid‑modified polymers.
- The TCM⁻ anion provides stable charge distribution that enhances surface conductivity without compromising polymer integrity.
Please contact us if you want to learn more or need assistance with your order